 Enlarge
Enlarge
British Mineralogy
Sulphate of Iron
- Class 3. Metals.
- Ord. 1. Homogeneous.
- Gen. 7. Iron.
- Spec. 6. Sulphate of Iron.
- Div. 1. Crystallized.
- Spec. Char. Sulphuric acid combined with iron.
- Syn.
- Vitriol martial. De Lisle, v. 1. 331.
- Sulfate de fer. De Born. v. 2. 39.
- Vitriol vert. Daubenton, 28.
- Vitriol of iron. Kirw. v. 2. 20.
- Fer sulfaté. Haüy, v. 4. 122.
- Vitriolum martis. Linn Syst. ed. 12. v. 3. 104.
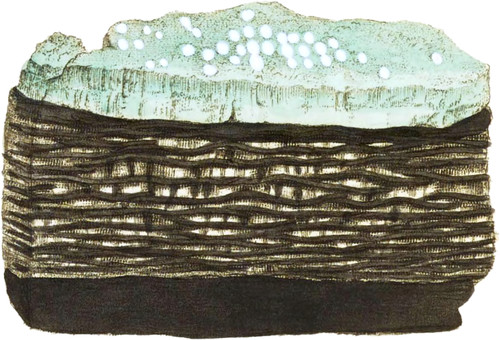
Green vitriol, as it is commonly called, is found crystallized, stalactitical, or in amorphous lumps, in many parts of Great Britain. The present is a curious specimen from Hawkshead coal mine, near Glasgow. It appears by a note sent to Mr. Vansittart with this and some other specimens which I had the pleasure of receiving from the Rev. Dr. Beeke, that the mine had been worked for above 200 years, from the crop to the dip (as the colliers term it), that is, following the descent from where it appeared on the surface, always working at the lowest part. Thus the upper parts, or pits, first worked were necessarily kept free from water, and were left exposed to the external air above the coal stratum. The black clay, or aluminous ore, being the cieling of the mine, absorbed the oxygen in the common air by means of the sulphure of iron, (which is almost imperceptibly mixed with it,) in such abundance as to expand it, first in the form of white silky threads, merely separating the laminæ in a somewhat undulating form, but afterwards expanding it in such a manner, that the whole stratum, which was but 14 inches, sometimes become a yard in thickness falling to the floor; and the threads, from being scarcely perceptible, become near an inch long, curling in many fanciful directions*. It sometimes ripens or consolidates into what the workmen call native copperas, and may possibly hold a little copper. it is somewhat crystallized, like the green part figured, upon the clay or alumine, which is in the act of throwing out little white opaque round spots, the effect of a further change since the specimen was in my possession. These probably contain less water than the other parts.—Its transparency is 2 or 3. Kirw. This is a very good alum ore, the sulphuric acid being by calcination separated from the Iron, and combined with the Argil, and an Alkali to form that substance.
- * Which will be shown in Plate xxviii.

