 Enlarge
Enlarge
British Mineralogy
Phosphate* of Lime
- Class 2. Earths.
- Order 1. Homogeneous.
- Gen. 3. Calx.
- Spec. 3. Phosphata.
- Div. 1. Crystallized.
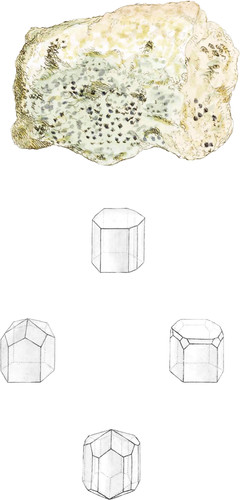
This specimen shows some of the varieties in the truncations and bevillings of the hexaëdral prism, from the alternately truncated edges—see the upper geometrical figure—to the more complicated—see the lower figure.
The gangue is commonly talcose, with Oxide of Tin and with Quartz, sometimes with Fluor, Feldspar, and rarely white Topaz, as in this specimen.
The prevalent quantity of Talc often gives it a waxy appearance, and this is peculiar to this rock, which, thus differing from the general appearance of rocks, has been supposed to he, and even sold for, Apatite itself.
Apatite has been confounded with Beryl by the Chemist Trommsdorf, who thought it to contain a new earth, which he named Augustite, but this is now understood to be an error. The upper geometrical figure shows the prism truncated on the three alternate vertical edges. The left hand figure below shows an hexaëdral pyramid placed on the solid angles of the prism. The right hand figure shows facets that are on the horizontal edges and solid angles. The lower figure shows both ends, forming the alternating pyramid, giving four- and six- sided facets to the prism†.
- * Phosphorus is sometimes united artificially with Lime, forming a powder, which, if put into water, sinks to the bottom, but soon decomposes the water taking up part of its oxygen, and freeing part of the hydrogen: the phosphorus becomes inflamed; and while this decomposition is taking place, flames burst forth through the water till the phosphorus is consumed.
- † It is remarkable that the pyramids in our crystals alternate with the faces of the prism. Haüy found them only opposite.

