 Enlarge
Enlarge
British Mineralogy
Magnesian Carbonate of Lime with Iron
- Div. 1. Crystallized.
- Syn. Sidero-Calcite. Kirw. 1. 105.
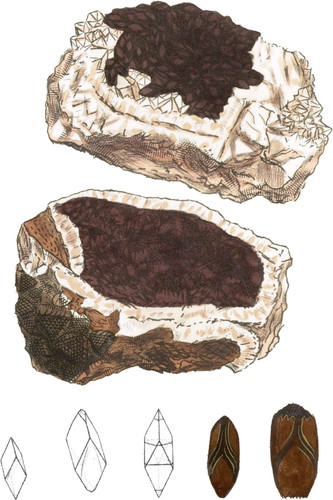
This curious Mineral was long since in Mr. W. Day’s collection, and I have had specimens sent me by P. Rashleigh, Esq. from Tincroft in the parish of St. John, Cornwall, as well as by another friend from that neighbourhood. All are related to each other in the form of their crystals, and instruct us in a series of modifications upon an acute rhomb; showing some very distinctly, and the manner of accumulation necessary to form others: a very natural lesson in Crystallography. In the upper figure are some minute perfect rhombs, resembling Haüy’s contrastante var. of Carbonate of Lime—see the bottom left hand figure; while others are larger with the three faces of the primitive remaining on the acute points or pyramids.
The middle figure shows a small truncation on the apex of the rhomb, forming a triangular facet, which in some Crystals is enlarged so as to cut off the obtuse lateral angles, thus producing hexangular faces at each end. The next magnified figure to that on the right hand shows art accumulation of flat or plated rhombs, forming truncations upon all the edges. The other figure shows them produced very widely, with the sharper angles accumulating in a triangle at both ends. This is exemplified in the lower part of the specimen fig. 2. The Crystal in the middle geometrical figure is also on a large specimen that was in Mr. W. Day’s collection.
This is the Braunspath of the Germans; but has, however, been sometimes taken for a Tin Ore in Cornwall.
Mr. W. Day observed that it did not effervesce with acids. It is one of its characters, as a Magnesian Iron Ore, to effervesce very slowly.

