 Enlarge
Enlarge
Exotic Mineralogy
Sulphuret of Mercury. Cinnabar
- Syn.
- Mércure sulfuré. Haüy 3. 437. Tab. comp. 78.
- Zinnober. Emmerl. 2. 143.
- Cinabre natif, oxide de Mércure sulfuré rouge. De Born. 2. 388.
- Native Cinnabar. Kirwan 2. 228.
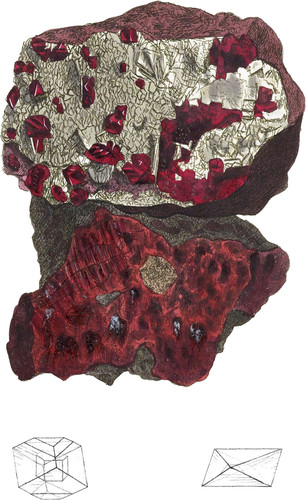
We are obliged to the Royal Institution for the use of this fine specimen, which was procured from Spain, along with the last, by the zealous I. G. Children, Esq. and the laudable desire of giving every help to facilitate public knowledge, made it even a pleasant obligation, I add part of the MS. account of the place from the valuable paper, of which I was allowed the use.
“The Quicksilver mines of Almaden are situated to the S. S. W. of Madrid, distant about 41 Spanish leagues, or about 164 miles English. The mountains in which they occur form part of the great Sierra Morena, and are principally Grawacke (coarse sandstone cemented by clay-slate, with more or less massy rolled stones of quartz, &c. generally most rich in ores). The vein stone at Almaden is of a firm compact crystalline texture, but at Almadenejos the Cinnabar is usually found in a dark coloured schist, soft and friable. The two mines are distant from each other about two Spanish leagues.
“The mines at Almaden are the oldest in the world; those at Almadenejos were worked many centuries since, but for some reason closed again; but in 1794 they were opened afresh, and are now extremely productive.”
Native Mercury is found in the greatest quantities at the latter place, and the finest crystallized specimens of Cinnabar, from one of which the present figure is taken. The series at the Institution are truly valuable, and instructive to consult. We herewith have the pleasure of expressing the wood-like, or fibrous Cinnabar, from the same place, and by the same friendly allowance. See the lower specimen.
The crystals are often externally splendent, and sometimes shining internally. Fracture conchoidal, also passing to powdery: Streak carmine red, or vermillion red, according to the density of the crystals. Volatilizes by the flame urged by a blow- pipe, giving the odour of sulphur, with a blue flame, and much smoke. Spec. Grav. from 6.902 to 8.160.
| Mercury | 84.50 |
| Sulphur | 14.75 |
| Loss | 75 |
| 100.00 |
Thus Mercury is changed, by about 171/2 per cent, of Sulphur, into Cinnabar.
The Hepatic ore is only amorphous, and more adulterated, having been found, upon analysis by Klaproth, to contain
| Mercury | 81.80 |
| Sulphur | 13.75 |
| Charcoal | 2.30 |
| Silica | 0.65 |
| Alumina | 0.55 |
| Iron | 0.20 |
| Copper | 0.02 |
| Water | 0.73 |
| 100.00 |
Cinnabar, artificially procured, is the usual pigment called Vermillion (scarlet), and it appears that the artificial compound is more durable than the native substance would be, when ground. The varieties of tint in the native Cinnabar may be owing to the nature of the solid while crystallized, as the powder would vary much less; and if it is not digressing too much, it may be compared to the darker octaëdral oxides of Copper, B. M, 53. 100, and 106, which are almost black with a metallic lustre, or Carmine or lighter red, but on powdering would produce indiscriminately a dull red.
Native Quicksilver is in globules among these specimens, which will be sufficient with the following description, if not found in Great Britain, and if it be, we shall be glad to shew it, not doubting but that it will be acceptable to our friends.
Quicksilver, or Mercury, was anciently known. It is solid at a temperature of 39° below Zero, and is said to crystallize in octaëdrons, it is then malleable, and contracts much, but at any heat above that point it is fluid; of course it is seldom seen in that state, but always fluid, in which it differs from every other native metallic substance. Its silvery lustre, and the particular freedom of motion when in the fluid stale, make it universally known. The drops divide into infinitely small particles, which are perfectly round, the larger are flat with obtuse edges, and a large quantity resembles melted lead in the obtuse or rounding edge. It easily divides, and as easily coalesces. It does not stick to the fingers, but is cold to the touch. It volatilizes after boiling at a heat of 656°. It is heavy, Spec. Grav. 13.581. It is often found occuring with Cinnabar, and is commonly wrought from it. It is of much use and consequence in Chemistry, the Arts, Mechanics, &c. Amalgamating with gold or silver, it is used to separate them from other ores, where fuel is scarce. With tin foil it forms our Mirrors, and is well known m Barometers and Thermometers, It is considered of much power in various combinations as a medicine.

