 Enlarge
Enlarge
Exotic Mineralogy
Aluminate of Lead
- Syn.
- Plomb-gomme (of the French.) Berzelius Nouveau Système de Minéralogie, ed. 1819, 283.
- Aluminate of Lead. Thompson’s Annals of Phil. XIII, 381.
- Native Hydrous Aluminate of Lead, James Smithson, Esq. in Annals of Phil. XIV. 31.
- Hydrate d’Alumine et de Plomb, M. de Laumont, in ib.
- Sel acide-phosphorique martial &c. Extrait d’un Mémoire sur la description de plusiers filons metallique de Bretagne, par M. de Laumont. Journal de Phys. XXVIII. 385. F 16.
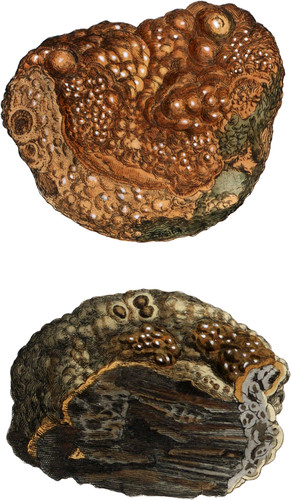
Plomb-gomme, as this mineral has lately been called, in consequence of its resemblance to Gum as it issues from the tree, does not appear to have been described by any author although it was mentioned as a peculiar mineral so long ago as in 1786 in the Journal de Physique by M. de Laumont, and even a few years previously by De Lisle: if his “Plomb rouge en stalactites” which he describes of a lively red, not orange or yellow colour, be the same substance. (Vol. III p. 399.) It has been found only in one place in the mine of Huelgoat, near Poullaouen, in Britany.
It occurs in a mamillated form encrusting the surfaces of hollows among stalactitical prismatic Pyrites, (decomposing Iron Pyrites.) It is of a bright wine yellow colour perfectly transparent, and possesses a high degree of lustre, its structure is like that of Malachite, and consists of numerous concentric layers formed of perpendicular fibres, the surface of the outermost layer is dull, but that of every internal one is highly polished and very brilliant; the hardness is superior to that of Fluor, but it yields easily to the knife; its specific gravity taken from a small piece which had several fissures in it was above 3.365. Under the action of the blowpipe it decrepitates violently; if exposed gradually to heat it becomes opaque, white, and contracts slowly in bulk, but docs not melt at any temperature, nor is it altered by charcoal; it effervesces rapidly when boiled in dilute Sulphuric acid, which dissolves the Argilla, and leaves Sulphate of Lead reducible upon charcoal; when heated below redness its water is given out silently. It was ascertained by the much to be regretted and unfortunate Smithson Tennant, Esq. when last in Paris, to be a compound of Oxide of Lead, Argilla and Water; since then Berzelius has made an accurate analysis of it, which is as follows.—
| Oxide of Lead | 40.14 |
| Argilla | 37.00 |
| Water | 18.80 |
| Sulphurous acid | 0.20 |
| Lime, oxides of Manganese and Iron | 1.80 |
| Silex | 0.60 |
| 98.54 |
Berzelius supposes the Sulphurous acid to be combined with parts of the Lead and Argilla forming Sulphites and with the Silex, Lime, &c. as accidentally mixed.
The acid stiptic taste mentioned by M. de Laumont probably arose from an admixture of Sulphate of Iron produced by the decomposition of its matrix.

