 Enlarge
Enlarge
British Mineralogy
Sulphate of Iron
- Div. 2. Imitative.
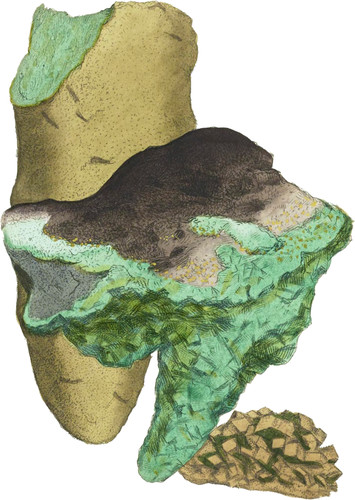
The upper specimen is a remarkably fine Stalactite from Cornwall, and has been in my possession many years without having apparently undergone any alteration, which is seldom the case. I have shown specimens in Plates 23 and 28 which have a very different appearance. It is commonly called Green Martial Vitriol, is usually soluble in twice its weight of cold water, and less than its weight of boiling water. It is said to have a double refracting power in proper and transparent Crystals. It is used for making ink with oak-galls, and gives it a peculiar astringent taste. It produces black with any vegetable, and is a styptic. The specimen figured above has some signs of the primitive rhomboidal angles about it.
The lower figure is taken from a North Wales specimen: it is more apt to decompose, and shows the changes it undergoes under these circumstances, as it becomes yellow or brown, sometimes crumbling into a Sulphur yellow. The primitive Crystal is somewhat apparent about this specimen; but the lowest figure is most perfect in that respect; such is sometimes found in Cornwall and Derbyshire, Dr. Kidd mentions finding it in the Shale that overhangs Odin mine at Castleton in the latter county. Sulphate of Iron is often of a fair green colour, (see Elucidation of Colours) lighter or darker, by which it is known from Zinc, which is nearly white, unless coloured a little with Iron or Copper. Sulphate of Copper is always much bluer, and approaches the Sulphate of Iron, if adulterated by Tron, which often happens. Spec. Grav. of Sulphate of Iron 1.83.
Analysis according to Kirwan:
| Oxide of Iron | 28 |
| Water | 46 |
| Sulphuric Acid | 26 |
| 100 |

