Introduction
Mineralogy is that part of natural science which treats of the unmixed, inorganic bodies occurring in nature, whether these be simple or compound.
It may, under a certain point of view, be considered as a department or offshoot of chemistry, but is nevertheless as much entitled to a separate place among the sciences as geology or botany. It must indeed be treated of independently, since by determining the differences of individual minerals, and by ascertaining certain common characters, it brings the variety of inorganic nature into a position for proper appreciation; just as is done for the organic world by its two sisters, botany and geology. As occupied with inorganic matter, it stands in closest approximation to geology, this treating of the combination of mineral matter into rocks, and the distribution, stratification, degradation, or decomposition of these rocks, in various ways and in different parts of the globe. Mineralogy deals only with individual specimens from these various rocks, which it classifies according to certain systems, describes them according to their external and internal characters, and adds to this the assistance furnished by chemistry, in determining their atomic constitution.
The expression, “unmixed, simple, or compound inorganic bodies,” requires some further explanation. The surface of the earth rarely affords the matter of its solids, its water, and its atmosphere, in an elementary, and consequently inseparable form. Indecomposable substances must be considered as simple matter furnished by nature. By far the greater number of mineral bodies are, however, not simple substances, but combinations of two or more such elements, in definite proportions, and in most cases even of a definite external form. Such compound bodies, to whose true character chemistry furnishes the only clue, are known as definite chemical combinations On the surface of our earth, for ages exposed to the alternating influence of volcanic heat, of water, and of oxydation, we find these definite chemical combinations of elementary matter, united in the most varied manner and proportion, by mechanical agencies. These mixtures, then, which exhibit no definite proportion, by weight, of the different constituents, are excluded from the department of mineralogy. From this statement, we may more clearly see the intimate connexion between chemistry, mineralogy, and geology, as already hinted at. When we separate mineralogy from chemistry, which in its system must, summarily at least, treat of all the combinations of matter, and consequently of all minerals, we must consider the analytical portion of the latter science as the instrument by which we construct the former on a sure basis. This indeed is the view which we have taken in the following pages.
![]() Testing of Minerals, and the Apparatus Necessary
Testing of Minerals, and the Apparatus Necessary
The chemical analysis of a mineral is indispensable to a proper knowledge of it. This analysis once performed, however, it is not necessary to repeat it in every case; all that is needed being to determine, with certainty, one or more constituents. The blowpipe (pl. 32, fig. 1) serves this purpose. It consists of a tube through which air is blown from the mouth into an expansion of the instrument, where it is somewhat condensed; thence to emerge with some force, through a small aperture at the extremity of a fine lateral tube. On bringing this aperture of the blowpipe over the burning wick of a spirit or oil lamp, the fine current produced as just described, causes the flame to run out into an attenuated point (fig. 2) of extraordinary intensity of heat; this flame is the agent employed in conducting the investigations. Mineral bodies, to be tested by means of the blowpipe, are placed in very small quantity on a piece of charcoal or clay, or else held in a platinum spoon or forceps (fig. 3), and thus submitted to the flame. This consists of two cones, one within the other; the former being blue, the latter yellow. The greatest intensity of heat is found just beyond the end of the blue flame. The blue flame is called the flame of reduction, bodies being deoxydized in it; the yellow is the oxydizing flame, causing, in many cases, the combination of oxygen with the bodies in question. Thus we may learn the melting point of bodies, or their relation to heat; we may reduce an oxydized body to its base in the blue flame, and oxydize it, if at all possible, in the yellow. By the application of such acids as phosphoric or boracic, capable of resisting the decomposing action of the flame, we dissolve a small portion of the mineral in the flame, and thus ascertain the color of its various salts. Another construction of the blowpipe is given in pl. 32, fig. 4. Here it is screwed to a post with an attached stand, and can be set higher or lower along the post, for the sake of allowing a proper position with reference to the accompanying lamp. It thus permits the free use of both hands.
A physical examination must frequently be combined with the chemical in the determination of a mineral. Thus the knowledge of its specific gravity is of great importance. In addition to the ordinary methods which may be employed, Nicholson’s areometer (pl. 32, fig. 5) is often used to great advantage. This consists essentially of the funnel a, the cylinder b, the rod cm, and the table or plate d. The instrument is so arranged that when set in distilled water, and a definite weight laid upon d, it will sink to a mark, m, made on the rod. To determine the specific gravity of a mineral it is laid on the plate d, when it will, of course, depress the instrument in the water. Additional weights must be added to bring the mark m to the level of the water, and the amount of these weights subtracted from the standard weight already referred to, will be the weight of the mineral in the air. Call this weighty. Remove the mineral from the plate and place it in the funnel or hollow cone a; immersed in the water the areometer will not sink quite to m, but about to c, the body losing in water an amount of weight equal to that of a quantity of water of precisely the same volume with itself, or equal to that of the water displaced. Additional weights are now to be laid on d until the level m is again reached: this amount, which we will call p′, expresses the weight of an equal volume of water. We have thus ascertained the weight of precisely equal volumes of water and of our mineral; and as water is the standard taken, \({\Large \frac{p}{p'}}\) will express the ratio of the two, or the specific gravity of the body. Thus x : 1 :: p : p′, and \(x={\Large \frac{p}{p'}}\).
The mineral must also be subjected to the tests afforded by electricity and magnetism. A simple instrument for ascertaining whether a mineral becomes electrical or not by friction, is represented in pl. 32, fig. 7b. Its principal part consists of a horizontally suspended insulated metallic needle. Whenever a body becoming electric by friction is presented to the needle, this, if in its natural condition, will be attracted: on the contrary, no effect will be produced when the body is a conductor. Fig. 7a is an instrument of similar purpose. At one end is placed a piece of tourmaline. On compressing the extremity of this between the fingers, the heat decomposes its neutral electricity, the positive passing over to the opposite extremity, and thus affecting the electrical state of the needles. The latter is insulated by a glass leg. On bringing any electric body near to the end of the needle, this will be attracted if the electricity be negative or resinous, and repelled if it be positive or vitreous. We are thus enabled to detect, not only the existence of free electricity in a body, but also its character. Pl. 32, fig. 6, presents a simple arrangement for determining the magnetism of a mineral. Here a is a magnetic needle, supported on the point of the pivot b, and, when permitted to play freely, taking up a position in the magnetic meridian. On bringing a magnetic mineral near to the poles of the needle, this will either be attracted or repelled, as the end of the needle to which the mineral is presented is of unlike or like character with the corresponding part of the latter. Should there be no free magnetism in the mineral, there will of course be no deflection of the needle.
The sensible internal peculiarities of minerals are very numerous. Thus a fresh fracture may be produced, and the color and degree of translucency or transparency of small chips observed; the color also of the powder may be compared with that of the solid mass. The shape of the natural fragments is also to be noted. Thus galena breaks into cubes, calcareous spar into rhombohedrons. and sulphuret of antimony into pointed crystals. In the natural cleavage of different minerals we observe a greater or less amount of roughness, of unctuosity or softness, and of polish. Some give out a peculiar sound when struck, or afford a peculiar taste, or smell, of which others again may be destitute. The determination of their hardness is of great value in distinguishing minerals, for which purpose we may employ the ingenious method of Mohs, of rubbing fragments of two different species together, and examining which of the two scratches the other. The different degrees of hardness may thus be represented by a scale beginning with the softest; and any mineral in the scale will be scratched by those below, but not above it. The hardness of any mineral may then be subjected to comparison with that of the standard. This scale of hardness, as introduced by Mohs, is as follows:
- 1. Talc; common laminated green variety.
- 2. Uncrystallized gypsum.
- 2\(\frac{1}{2}\). Foliated mica.
- 3. Transparent calcareous spar.
- 4. Crystallized fluor spar.
- 5. Apatite.
- 5\(\frac{1}{2}\). Scapolite-crystalline.
- 6. Feldspar-white, cleavable variety.
- 7. Transparent quartz.
- 8. Transparent topaz.
- 9. Corundum (sapphire).
- 10. Diamond.
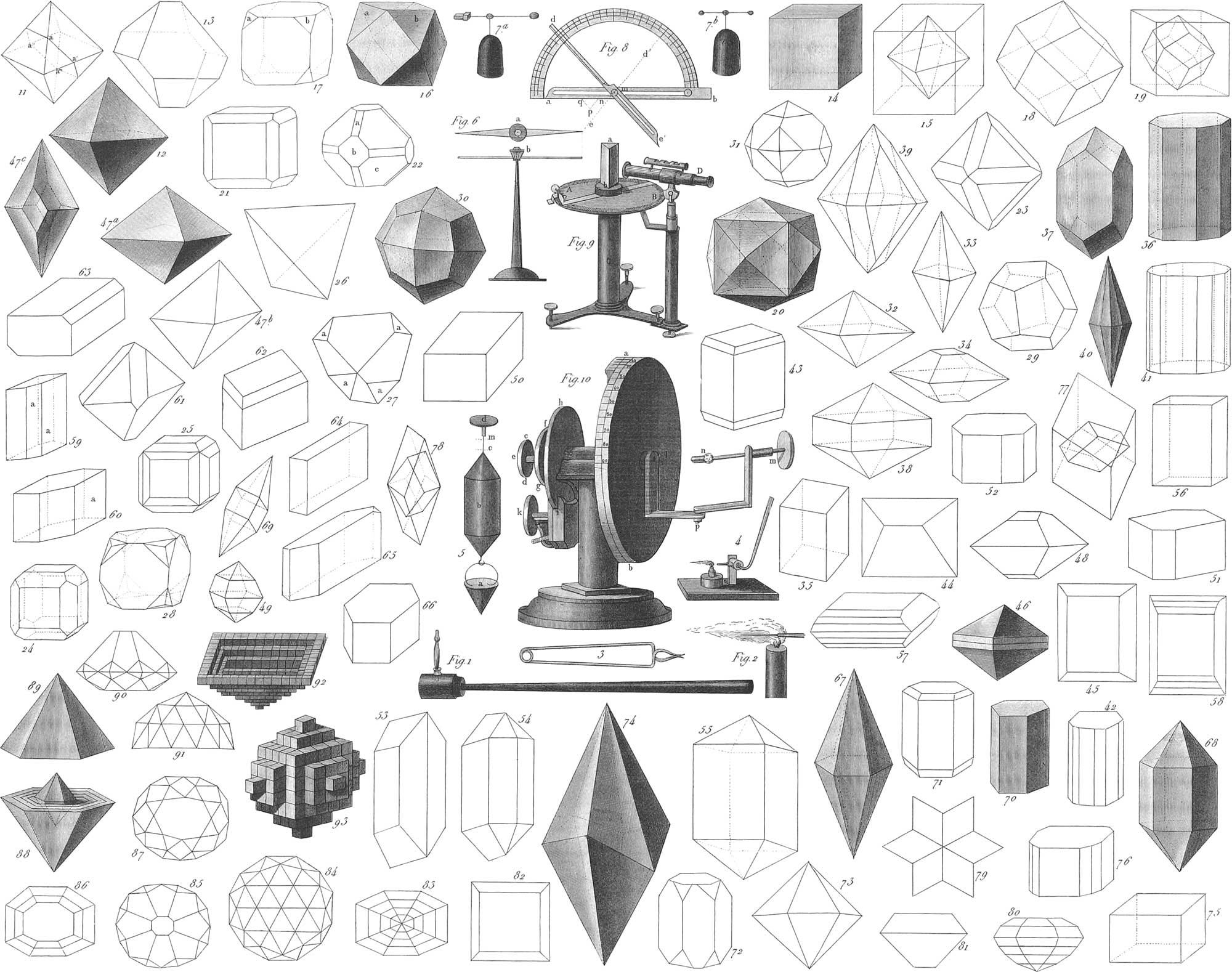
Thus, a mineral which abrades feldspar, but not topaz, is said to have a hardness of seven, equivalent to that of transparent quartz, &c. The externa] shape of minerals is also of great interest and importance. This, indeed, in many cases is objective, being determined by that of other bodies; sometimes by the gravity of the mass itself; but in most instances there is another subjective form, independent of extraneous influences, and peculiar to the particular species of mineral. Direct observation first of all teacher us that the natural form of every mineral is a solid, bounded by plane surfaces. A still closer examination enables us to ascertain that the true external shape of a mineral is that of a closed figure, bounded by sharp edges and angles, the points and lines of intersection of the planes just mentioned. Bodies thus inclosed are called crystals. In view of the apparently infinite variety of crystalline forms, we might at first be induced to suppose that the precise outline of any body is capable of being infinitely and indefinitely varied; observation, however, shows this not to be the case, nature here exhibiting a remarkable simplicity, and a most admirable law. As mineralogy deals with solid bodies, that method for their determination were most desirable which mathematics shows to be the simplest. A body has height, thickness, and breadth; thus, three dimensions. Clearly to illustrate and compare these three forms of extension, let us suppose the height to be indicated by a vertical line, which shall in future be called the principal axis. Let other lines be passed through this principal axis, at right angles to it and to each other, these lines to be called the secondary axes. These three axes measure the three dimensions of a body, and it will be found on examining these dimensions that the natural distances between the surfaces bounding the crystals are not without a rule, but are rather determined by laws referable to variations in the ratio of the length and angular relations of these axes. According: to various definite ratios between the three axes, we may group a large number of, at first, apparently different crystalline forms. However different the ratio of three such lines may be in respect to their length, estimated from their middle, or point of intersection, or however varied the angle of inclination, nevertheless we know of but six essentially different sets of axial proportions. The simplest figures determined directly by these axes are called primary forms. All the primary forms, with the secondary forms derived from them, however different they may seem, are referable to one of six systems of crystallization.
![]() Crystallography
Crystallography
That part of mineralogy relating to systems of crystallization, is called Crystallography. The systems referred to in the preceding paragraph are briefly as follows:
I. The Monometric System, Dana. The Regular System (Das regulaire System), Weiss. Tessular, Mohs. Tesseral, Naumann. Isometric, Hausmann.
The character of this system is such that, if a certain point be taken; and three axes be drawn through this point, at right angles to each other, they will all be bounded at equal distances by a solid angle, a face, or an edge. It is therefore a matter of indifference which axis we make the vertical or primary, all three being of equal value. The regular octahedron is generally taken as the type of this system, the others being derived from it. The most general forms are as follows:
1. The regular octahedron (pl. 32, figs. 11 and 12). This is a solid inclosed by eight equilateral triangular faces, intersecting each other, in six solid angles, and twelve edges. Connecting each opposite pair of solid angles will give us the three axes, intersecting each other at right angles in a common point, and of the same length from the point of intersection. Whatever two opposite angles be selected as the limits of the vertical axis, the others will always have the same relative situation. Natural crystals, however, do not always exhibit the perfect symmetry thus indicated. Distortions frequently occur, one of which is given hereafter as a derivative of the octahedron. Thus if we intersect any two parallel faces of this solid, by a plane parallel to another face, or if we move one face parallel to itself, nearer to the centre, a hexagon (fig. 11, a, a′, a″, a‴), will be obtained by the lines of intersection.
By moving two parallel faces towards the centre, these with the six other abbreviated faces will inclose an octahedron abbreviated to a six-sided plate (fig. 13). Octahedrons distorted in this manner are more abundantly found than absolutely symmetrical octahedrons: it must nevertheless be observed, that such distortions are always produced by some external impediment, and that nature, in the entire absence of all obstructing influences, always exhibits perfect symmetry. This is true, not for octahedrons alone, but for all crystals.
2. The cube (fig. 14) is produced from the octahedron, by truncating its solid angles by planes perpendicular to the axes. Here the axes are again all equal, and connect the centres of opposite faces. Fig. 15 shows the relation of the cube to the octahedron.
3. The cubic octahedron (figs. 16 and 17) is obtained from the cube, by truncating the six corners, until the old faces again become squares. a and b are new faces parallel to the old faces of the obliterated octahedron.
4. The rhombic dodecahedron (pl. 32, fig. 18) is produced from the cube by the truncation of its edges, until the original faces are obliterated. This solid has twelve rhombic faces, twenty-four equal edges, and fourteen solid angles. Of these solid angles, six are formed, each by four rhombs meeting by their acute angles; and eight, each by three rhombs meeting by their obtuse angles. The relation of the rhombic dodecahedron to the cube is shown in fig. 19.
5. The pyramidal cube, or tetrahexahedron (fig. 20), is produced by placing a four-sided pyramid on each face of the cube.
The cubic octahedron has already shown that a crystal may be inclosed by faces belonging to two different forms of crystals.. This case is often repeated. Thus, in figs. 21 and 24, the cube is represented with dodecahedral faces replacing its edges. Fig. 22 shows an octahedron, with dodecahedral faces, a, and cube faces, b; c, indicating what is left of the octahedron. Fig. 23 represents an octahedron passing into a dodecahedron: fig. 25 is a combination of cube faces and those of the pyramidal cube; or a cube with its edges bevelled. Fig. 30 is the trapezohedron, or tetragonal trisoctahedron; a solid bounded by twenty-four equal trapezia. It can be derived from the octahedron by replacing its corners by four faces, or by replacing each corner of the cube by three faces (fig. 28).
The figures hitherto derived from the primary forms have been produced by modifying all the similar parts of the primary simultaneously. Such forms are called holohedral. Hemihedral forms of crystals occur in equal number. These are forms in which half of the similar parts of the crystals are modified alike, independently of the other half. Some of these forms are:
6. The tetrahedron (fig. 26), a solid, inclosed by four equilateral triangles. Fig. 27 represents an octahedron passing into a tetrahedron, in which the faces, a, indicate what is left of the octahedron faces. A form of frequent occurrence, and likewise belonging to this place is:
7. The pentagonal dodecahedron, or hemi-tetrahexahedron (fig. 29). This is a hemihedral form derived from the pyramidal cube, and bounded by twelve equal pentagons.
We may remark, in reference to this as well as other systems, that all the different forms belonging to one and the same system, may occur in the same mineral. Thus iron pyrites or sulphuret of iron occurs in cubes, octahedrons, pentagonal dodecahedrons, rhombic dodecahedrons: frequently in the same crystal we may have faces of all these different forms.
II. The Dimetric System, Dana. Das viergliederige System, Weiss. Pyramidal, Mohs. Tetragonal, Naumann. Monodimetric, Hausmann.
The fundamental form of this system is an octahedron with a longer or shorter vertical axis and two equal lateral axes (the three axes all at right angles to each other).
1. The Square Octahedron. It is called obtuse (fig. 32) when the vertical axis is shorter than the other, and acute (fig. 33) when longer.
The figures of the derivative forms are based on these proportions of the primary axes. As the vertical axis is always unequal in respect to two equal lateral axes, there will be different values in the derivatives. Thus the two corners through which the primary axis passes (pl. 32, fig. 34) may be truncated without the four corners of the secondary axes requiring a similar truncation. Elongations and contractions may occur in the direction of the primary axis, while the two secondaries mutually retain an equality of length.
The acute, like the obtuse octahedron, has the same number of faces, edges, and corners, or solid angles, as the regular; the base is in all a square; the side triangles, however, bounding the solid, are in the former isosceles, in the latter equilateral. The derivative forms from the square octahedron are, the octahedron (fig. 39) with truncated lateral edges: fig. 38 with truncated, and fig. 46 with bevelled basal, edges. Also the octahedron with the corners of the vertical axis replaced by four plane faces (fig. 49).
2. The Right Square Prism (fig. 35). It is produced by truncating the four basal edges of the octahedron, as also the extremities of the vertical axis. The figure is bounded by four equal lateral rectangles and two terminal squares. Fig. 37 represents the right square prism, with its corners truncated until the terminal faces are replaced, each by a solid angle.
3. The Regular Eight-sided Prism (fig. 36) is obtained by bevelling the vertical edges of the right square prism. The double eight-sided pyramid (fig. 40) is produced by cutting off the basal edges of the eight-sided right prism (fig. 36), by planes meeting at the extremities of the vertical axis, and obliterating the lateral faces.
4. The Twelve-sided Prism (fig. 41) is produced by bevelling the vertical edges of the right square prism, without obliterating the lateral faces, as in the regular eight-sided prism.
5. The Square Plate, with Four Bevelled Edges (fig. 82), is produced by truncating the extremities of the square octahedron.
III. The Trimetric System, Dana. Das zwei-und-zweigliederige System, Weiss. Orthotype, Mohs. Rhombic, Naumann. Holohedral-rhomoic, Breithaupt.
The axes in this system are at right angles to each other, but all of unequal length. The fundamental form is:
1. The Rhombic Octahedron. If we suppose the square base of the preceding system to become oblique, a rhomb will be produced. If we suppose the lateral axes to occupy the place of diagonals to this rhomb, one will connect the acute and the other the obtuse angles, and while at right angles to each other, will be of unequal lengths. The vertical axis, passing through the point of intersection, and perpendicular to their plane, is either shorter than the lateral, in which case we have the obtuse rhombic octahedron (pl. 32, fig. 47a), or else more elongated, producing the acute rhombic octahedron (pl. 32, fig. 47c).
Since all three axes are unequal, it is a matter of indifference which we take as the principal or vertical axis. Having assumed one, those two corners alone have equal crystallographical value which lie on one and the same axis. Any two such corners may, for instance, be bevelled (fig. 48), without this modification needing to be extended to the other corners. The general shape of this form is that of the square octahedron, only the base here is rhomboidal. Thus the twelve edges of this octahedron are different in the whole of three different kinds. Derivative forms from this are:
2. The rectangular prism (fig. 56). It is produced by passing planes through the rhombic octahedron at right angles to the axes. Supposing these to increase until they intersect and truncate the two extremities of the primary axis, we shall obtain this figure. Sharpening the lateral faces of this prism, we shall obtain an octahedron (fig. 47b), whose base is a rectangle. Figs. 44, 45, 57, and 58, represent modifications of this octahedron.
3. The right rhombic prism, terminated by the faces of the rhombic octahedron (fig. 69). Truncating the four horizontal basal edges of the rhombic octahedron by planes parallel to the vertical axis, and letting them enlarge until they intersect, we shall have this figure. It may be considered as the passage of the rhombic octahedron to the following.
4. The right rhombic prism (fig. 50). This is produced by truncating the two solid angles of the vertical axis, and letting the new faces thus formed enlarge until they intersect the faces truncating the basal edges of the last figure, these also being supposed to be extended. The figure thus formed will be bounded by two rhombs and four rectangles.
5. The irregular six-sided prism (fig. 51) is formed from the preceding by truncating the two obtuse or the two acute lateral edges.
6. The irregular eight-sided prism (fig. 52) is produced when the acute and obtuse lateral edges are so replaced by faces as still to leave the original lateral faces.
7. Figs. 53, 54, and 55, present some combinations of prisms and octahedrons, belonging to this same system, and frequently observed in certain well known salts. Figs. 53 and 54 are crystals of saltpetre. Fig. 55 often occurs in sulphate of tin.
IV. The Monoclinic System, Dana. Das zwei-und-eingliederige System, Weiss. Heminorthotype, Mohs. Monoclinohedric, Naumann.
The axes by which we measure the dimensions of this system are two lateral, unequal, crossing each other at a right angle, and a third oblique to one of the lateral, but at right angles with the other. Considering an octahedron as belonging to this system, its upper and lower faces would be different. It is customary to consider an oblique rhombic prism as the primary form, whose extremities stand perpendicularly to the lateral edges, and at an oblique angle with reference to the other two. From this prism the other forms may be derived as before. Pl. 32, figs. 59 and 60, represent two prisms in which two edges are truncated by the face a, producing oblique six-sided prisms. Fig. 61 represents an octahedron with half the edges truncated. Fig. 62 is a prism corresponding to the case in which half the basal edges are truncated.
V. The Triclinic System, Dana. Das Ein-und-eingliederige System, Weiss. Anorthotype, Mohs. Triclinohedral, Naumann.
An oblique rhomboidal prism is the basis of this system. All three axes are here oblique and unequal. A prism of this character is shown in figs. 64 and 65. In this system only two parallel faces and two opposite edges are of like value. For this reason we see the truncation of the edges extended only to two diagonally opposite edges, as in fig. 63. The inclination of the plane which truncates these edges is different with respect to one face of the edge from the other; and the six-sided prism (fig. 66) is consequently irregular.
VI. The Hexagonal System. Das Sechsgliederige System, Weiss. Rhombohedral, Mohs. Hexagonal, Naumann, Dana. Monotrimetric, Hausmann.
This system exhibits a striking peculiarity, as compared with the others. While, in the preceding systems, the dimensions of bodies were given in the least number of axes (namely, three) in which their exteriors could be considered; the simplest conditions are obtained by assuming four axes. Three of them lie in one plane, and having equal inclination to each other, are of equal length: they thus form the diagonals of a regular hexagon. The fourth, assumed as the vertical axis, is unequal to the three others, and stands perpendicular to their plane. As the primary form of this system we may assume the double six-sided pyramid (figs. 67 and 73). This is a solid bounded by twelve isosceles triangles. Truncating the basal edges by planes parallel to the vertical axes, will give us the pyramidal six-sided prism (fig. 68). Truncating the terminal solid angles of fig. 68 will give us the regular six-sided prism, with right terminal faces (figs. 70 and 71). Fig. 72 is obtained by truncating the corners of fig. 70; bevelling the six vertical edges we have the twelve-sided prism (fig. 42); and by bevelling four edges the prism (fig. 76). A form of this system, occurring frequently in calcareous spar, is the scalene octahedron (fig. 74). The hemihedral shape of the double six-sided pyramid, or the rhombohedron (pl. 32, fig. 75), is often assumed as the primary form of this system. One and the same crystal is thus frequently inclosed by faces of several different rhombohedrons, derived from the fundamental rhombohedrons. Figs. 77 and 78 represent the natural situation of two rhombohedrons.
![]() Internal Structure of Crystals
Internal Structure of Crystals
What we have said in the preceding section has reference to the external form only, whatever might have been the relation in this respect between the different figures. A close investigation, however, discloses the fact, that they sustain remarkable relations in respect to their internal structure. Many crystals, on being broken, naturally separate into definite forms, as calcareous spar into rhombohedrons, and galena into cubes. Nearly all crystals may be split, by means of a sharp knife and a hammer, into certain other forms of the same system. Thus fluor spar may, by cleavage, be converted from a cube into a regular octahedron. We may, in many cases, therefore, suppose a crystal to be a mass built up of those elementary forms into which they split. Hauy, the founder of the science of crystallography, assumed an elementary form for each system, and supposed all forms belonging to a system as composed of aggregations of such simple forms. It would rather seem, however, that each crystal is composed of very small crystals of the same form; that a cube must be composed of smaller cubes; and that an octahedron is not. composed of regularly decreasing layers of small cubes, as according to figs. 92 and 93, given by Hauy, but consists of a mass of very small octahedrons. Besides these peculiarities, there are crystalline formations of interest, which appear to consist of a series of layers, regularly applied to an originally minute crystal. On making a section of such a crystal, we may see a series of concentric outlines inclosing the central nucleus, these different outlines indicating so many different layers, being often of different colors. This feature is often seen in six-sided prisms of calcareous spar, and in quartz (figs. 88 and 89). Besides the fact that a large crystal may be produced by a regular aggregation of smaller ones, several distinct crystals may unite or grow together, and produce a definite form. Combinations consisting of two individuals are called twin-crystals. Groups of a similar character, consisting of many individuals, are frequently found. Substances which crystallize in right rhombic prisms, sometimes exhibit a stellated grouping (fig. 79).
![]() Measurement of the Angles of Crystals and the Necessary Apparatus
Measurement of the Angles of Crystals and the Necessary Apparatus
In section two it has been shown that the positions of the surfaces inclosing a crystal depend on the axes. Hence it follows, that as these axes occupy a definite angular relation to each other, the faces of the crystal must mutually exhibit the same relation. In consequence of this dependence we are enabled to tell the inclination of the axes, knowing that of the faces, and consequently to determine the system. Certain instruments are, however, necessary, accurately to determine the precise angle made by two plane faces with each other.
The oldest and simplest instrument is the common goniometer (pl. 32, fig. 8). It is of easy use, and very convenient, when we do not require an angle with any very great accuracy. To determine an interfacial angle, one face must be applied to an, with the edge of inclination opposite to the centre of the diameter. The arm, e′d, is then to be brought around until its edge rests on the other face: the crystal is thus accurately inclosed between two radii of the semicircular scale. The angle indicated on the point of the scale, crossed by the prolonged movable radius or arm, indicates the angle of inclination desired, provided that the edge, ab, coincide accurately with the single diameter of the semicircle. This diameter is jointed at b, to admit of measuring angles which are partially imbedded. When the application of the two radii is completed, the joint, m, is to be tightened, and the whole brought back, until ab again coincides with the diameter. The inclination of the planes not intersecting each other in an edge, may also be measured by turning the movable arm until the two edges of the instrument are parallel to, or coincide with the plane faces in question.
Much more accurate measurements may be obtained by the use of Wollaston’s reflecting goniometer (pl. 32, fig. 10), an instrument recently very much improved. Here, ab is a disk turning on an axis, el, carrying a circle graduated to degrees on its periphery. Rotation takes place in such a manner that the disks, fg, hi, and ab, all turn at the same time with the horizontal cylindrical body forming their axis y in this axis another axis, el, may itself be turned by means of the button, cd; k is a clamp-screw, by means of which the entire system, fg, hi, ab, may be fixed by tightening hi; at the same time that this is done, the inner axis, el, may still be turned independently of the button cd, and any required position thus be given to the arm lp. This arm carries the jointed continuation, pm, turning on p. At n the crystal to be measured is fixed, and so adjusted, that the edge in which the two planes (whose inclination is to be ascertained) intersect shall be parallel to the axis el. The circle, ab, is now fixed by means of the screw k, and the axis el, turned by means of the button cd, so that the image of some distant object, as the horizontal bar of an opposite window, may be seen in one of the planes of the crystal. The spot from which the image is seen in the face of the crystal is marked by drawing a black line on paper, and so placing this paper as that its line may be seen as a continuation of the reflected line of the window-bar in the crystal. If we were to suppose a thin metal plate, polished on both sides, to be fixed at n. instead of the crystal, after obtaining the image of the window-bar in one of the faces, as just described, we should have to rotate it just 180° to see the same image in the other face. The case is similar in respect to our two crystal faces. If two of these be parallel, like the faces of our plate, the angle thus obtained will be 180°. Any other inclination will be obtained in a similar manner. The operation must be commenced by bringing 180° on the disk directly opposite to the 0° of the vernier a, and establishing a perfect parallelism between the edge of our crystal and the standard window-bar. The image of the window-bar, seen at this time, must be on a line with the black line drawn on the paper already referred to. The whole apparatus, clamped together, is next to be turned until the same bar is seen in the other face, and on a line with the line on the paper. The angle through which the apparatus has been rotated, as read off on the vernier, will be the angle of inclination of the two faces. For further remarks on the subject of the reflecting goniometer see Optics.
For very large crystals, in which the preceding form of reflecting goniometer cannot be used, we may advantageously employ the goniometer of Gambay (pl. 32, fig. 9). It consists principally of a horizontal and rotating disk, AB, supported by a stand, C. The crystal being fixed in the centre of the smaller disk, ab, the telescope, D, is now directed towards the crystal, and this turned until some distant object, as a distant tower, shall be seen in the cross-hair of the former. The angle through which the disk, ab, bearing the crystal, must be turned to see this same image crossed by the cross-hair in the other face of the crystal, will be the angle of inclination of the two faces.
![]() Special Mineralogy
Special Mineralogy
The succeeding descriptions of the minerals figured in plates 33, 34, 35, and 36, will allow of a brief survey of the application of crystallography; as also of the manner of mineralogical examinations. The number of single minerals already determined, with great exactness, is so great, that we can no more omit system in mineralogy, than we can in botany or zoology. The minerals figured in our plates have therefore been, to a certain extent, arranged according to the system of Hausmann, which is the one we have deemed best for our purpose.
We thus divide minerals into ten classes : 1, Metalloids; 2, Native Metals; 3, Tellurids; 4, Antimonids; 5, Arsenids; 6, Selenids; 7, Sulphurids; 8, Oxydes; 9, Silicates; 10, Salts.
Class 1. Metalloids.—Hausmann
Simple non-metallic bodies, which disappear when heated in the air or in oxygen, forming a combination with the latter.
1. Sulphur.
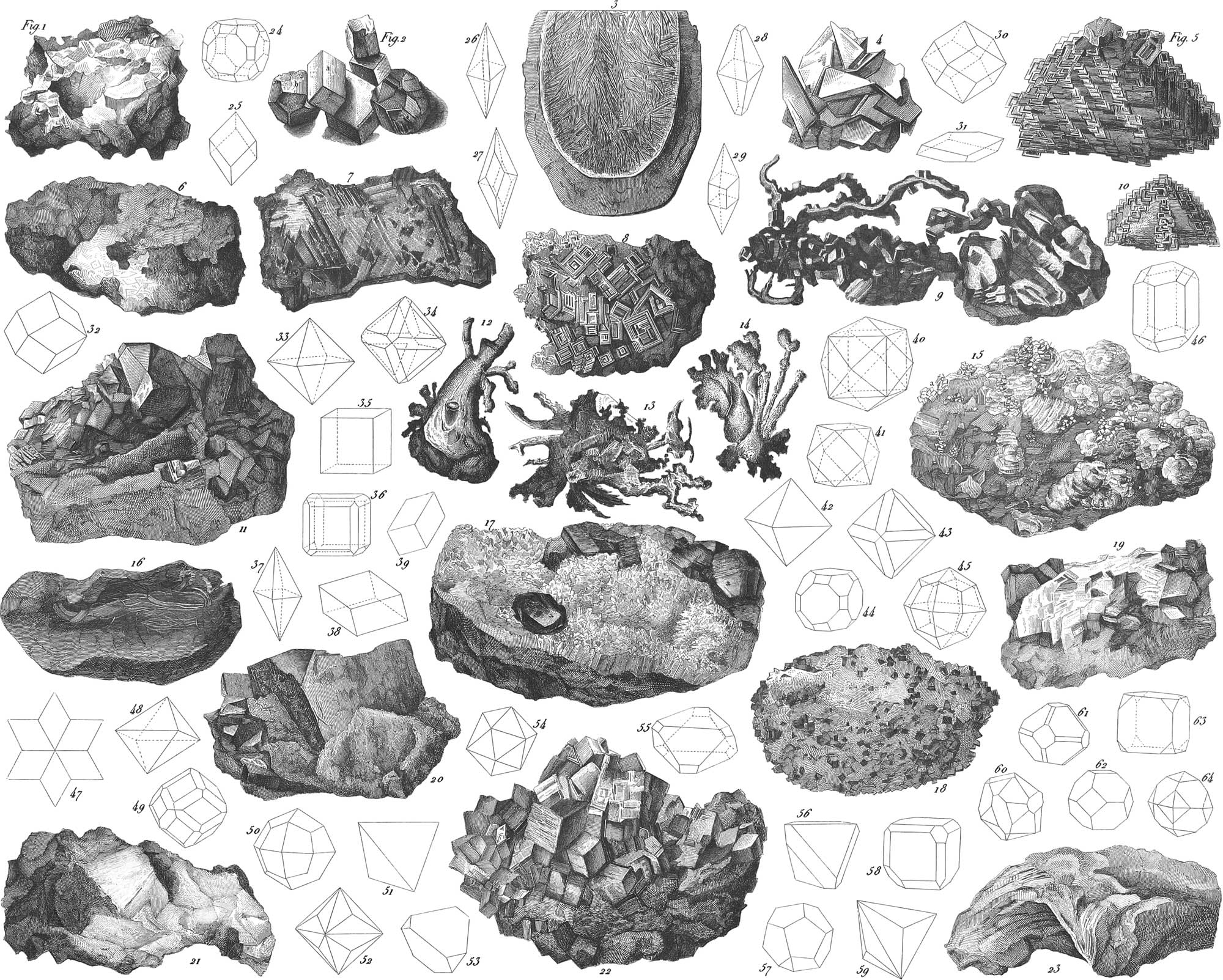
This is a substance occurring in great abundance. It is found both simple or native, and combined with other bodies. The combinations most generally met with are those with the metals, and with oxygen; less abundantly with hydrogen. It is frequently, although in small quantities, found as an essential ingredient in organic substances. Native sulphur is of a yellow color, and has a specific gravity of 2.07. It is a non-conductor of heat, and becomes negatively electric by friction. It is one of those substances whose crystals cannot all be referred to the same system; it is therefore dimorphous. The crystals of native sulphur are rhombic octahedrons (pl. 33, figs. 4, 26, 27, 28, and 29), whose sharp solid angles are frequently truncated, as in fig. 27. Those crystals, however, which shoot out in melted sulphur when cooling, belong to the monoclinic system; they appear generally in the shape of long, thin, and brittle needle-shaped crystals, as shown in fig. 3. Sulphur has a resinous lustre, is sub-translucent, and gives out a peculiar odor when rubbed. It readily melts by the application of heat, and then takes fire when exposed to the air, burning with the production of sulphurous acid gas, and a suffocating odor. Native sulphur is of frequent occurrence in different formations, but rarely in large quantities. The finest crystals are found in the valleys of Noto and Mazzaro, in Sicily. It is found in very large quantities about recent or extinct volcanoes, as in the Solfataro near Naples. Sulphur often occurs in an impure earthy condition in large masses, and is deposited from sulphur springs. Most of the sulphur of commerce is obtained from Sicily, or else is derived from the sulphurets. In combination with metals it is universally distributed, and in large quantities, and is obtained in the reduction of the metal as a secondary product. Its applications are very numerous, especially in the different forms of matches, gunpowder, &c.
2. Carbon
Carbon occurs in a naturally pure and crystallized state, under forms belonging to two different systems, namely, as diamond and as graphite. It likewise occurs in an amorphous condition as stone coal, impure by being mixed with the other ingredients forming the carbonized plant. The purest form of carbon, the diamond, exhibits a crystallization belonging to the regular system. The most common forms of the diamond are the regular octahedron (fig. 42), and the regular octahedron with the faces of the rhombic dodecahedron (fig. 43). It more rarely occurs in cubes, tetrahedrons (fig. 51), and trigonal-polyhedrons.
The diamond was first found in the East Indies. The most important diamond mines at present, are, in India, between Golconda and Masulipatam, in Brazil, and in Borneo and Malacca. Here they occur in alluvial soils, lying loose in the sands of plains, and in the beds of rivers, or in the ferruginous clay and recent conglomerate composed of quartz grains cemented by ferruginous sand. Nothing certain is known of the origin of the diamond. Of all gems this is the most esteemed, adding to its other properties that of being the hardest known. Its refractive power is extraordinary, and Newton, as early as 1675, concluded from this quality, peculiarly high in bodies containing carbon, that the diamond must be combustible. Its combustion was first effected by the Florentine Academy in 1694, and Lavoisier, by proving it to consist of pure carbon, first announced its true nature. It is a non-conductor of electricity, and has a specific gravity of 3.5. It occurs quite colorless, or else grey, brown, black, yellow, green, and blue. It is used very advantageously as a means of cutting glass; but its chief application is as an ornamental gem, after being ground in certain regular shapes, whose end is the production of the peculiar and sparkling lustre so much admired. Colorless diamonds are most esteemed as ornaments. The impure and small diamonds are reduced to powder to be used in grinding the rest. The grinding of the diamond requires great skill, and the different forms imparted have definite names. The principal of these are: 1, The brilliant (pl. 32, figs. 85, 87, 90); 2, The rosette or rose (figs. 84, 91). Older forms, now rarely imitated, are shown in figs. 80, 81, 82, 83, and 86.
The second form of carbon is graphite, known also as plumbago and blacklead. It also occurs so pure as to leave but little ash when burned. Certain characteristics distinguish it very decidedly from the diamond: it does not crystallize in the regular system, but in six-sided plates; is a good conductor of electricity; and has a low specific gravity, 2.14–2.27. Graphite is soft, unctuous to the touch, of a steel-grey metallic lustre, and opake. In thin plates it is flexible. It occurs in beds and layers in primitive rocks, gneiss, granite, primitive limestone, greywacke, and greenstone. The principal localities are in England, Germany, Norway, Greenland, France, Spain, the United States, &c. Its uses are various, being employed in fabricating pencils, in the construction of crucibles and small furnaces, as an anti-attritient, &c. The best graphite for the manufacture of lead pencils comes from the Borrowdale mine, England. The poorer graphite is extensively used, and, by pressure, good pencils are made.
The third kind of carbon, the uncrystallized, which occurs in various combinations with hydrogen and oxygen, will be discussed under the head of Geology.
Class 2. Native Metals
Simple bodies, or occurring as mixtures or alloys with one another, in variable or indefinite proportion; specific gravity from 7 to 22. All possess a metallic lustre, or at least acquire it under the burnisher. They are opake, and good conductors of electricity.
1. Native Iron
Iron rarely occurs in the native or uncombined condition. It is found as such in small masses or grains only in mica slate, and in large masses as meteoric iron. In its combinations with the other elements, and especially oxygen and sulphur, it is the most generally and abundantty diffused of all metals. Metallic iron was known in very ancient times, since Moses speaks of iron knives. With respect, however, to the manner in which the ancients obtained their iron, we know little or nothing, except as far as it may have been derived from aerolites. Instead of iron the ancient Greeks used a mixture of tin and copper. Meteoric iron sometimes occurs in large masses. One of these, found in South America, weighed nearly 40,000 lbs. Remarkable specimens, also, are found in the cabinets of St. Petersburg and Vienna. One in the cabinet of Yale College, at Newhaven, weighs 1635 lbs. However rare the fall of meteors may be, they are mentioned by as early an author as Pliny. The fact of their existence was subsequently doubted, until Chladni, in 1794, took up the question, and soon accumulated such a mass of proof as to satisfy the most sceptical. Meteoric iron generally occurs in jagged and roughly- vesiculated fragments (pl. 33, fig. 1). It is not perfectly pure, generally containing a slight quantity of some other metals, of which nickel has been found in all undoubted meteorites. In the meteoric iron of Ellbogen were found, in one hundred parts: iron, 88.231; nickel, 8.715; cobalt, 0.762; and manganese, 0.249. Meteoric iron has a crystalline texture by which it is recognisable. By sawing off a corner and polishing it, the application of dilute nitric acid will develope triangular and lozenge-shaped figures, mostly of 60–120° inclination, the sides all bounded by double lines. These figures, shown in fig. 7, are known as the Weidmannstedtian figures, from their discoverer. The iron of commerce is obtained in furnaces by reduction from oxydes or carbonates.
2. Native Silver
Silver frequently occurs native in silver mines, and also alloyed with other metals or simple bodies; in the form of sulphuret of silver it is very abundant. Native silver occurs crystallized (pl. 33, fig. 9), exhibiting the forms of the regular system, as the cube, the rhombic dodecahedron (fig. 30), the pyramidal cube (fig. 40), and the octahedron (fig. 42); or else it is found in filiform (fig. 16), or in arborescent (fig. 12) shapes, sometimes in coarse masses, in plates, &c. Native silver, like most native metals, is not perfectly pure, containing a variable amount of gold, copper, antimony, and arsenic, without any change being produced in its external or crystalline form. Its color alone is affected in these mixtures, varying from silver- white to brassy-yellow. Silver is very ductile and malleable, and difficult of fusion. It occurs mostly in veins, more seldom in beds in primitive rocks, granite, porphyry, and gneiss; in transition and stratified rocks, accompanied by barytes, hornstone, or calcareous spar. The principal localities of native silver are in Saxony, Bohemia, Hungary, the Hartz. Norway, Siberia, Mexico, &c. It occurs associated with the native copper of Lake Superior. The silver of commerce is obtained partly from native silver, partly from the sulphuret, or from combinations of the sulphuret with other sulphurets, such as antimony and arsenic. It is found in especial quantity in reducing lead from galena (sulphuret of lead). It is obtained by reduction, by amalgamation, or by cupellation.
3. Native Gold.
Gold rarely occurs in any other form than native. In this state, like silver, it is alloyed at times with other metals, in variable, not definite proportions. The principal alloys are with silver, copper, iron, platinum, iridium, and palladium, the crystalline form not being necessarily changed thereby. The color of these alloys varies from golden-yellow to silvery-white. It has a specific gravity of from twelve to twenty, is rather soft, and exceedingly extensible. The crystals of native gold belong to the regular system; the most common forms are shown in pl. 33, figs. 32, 33, 35, 36, 40, 41, 42, 43, 44, 45. It also occurs in various uncrystallized forms; in arborescent ramifications, as in fig. 14; also in filiform and reticulated masses, in coarse lumps, scales, and grains. Its original locality is in veins and laminæ in quartz, in talcose micaceous rocks, greenstone, and often associated with sulphuret of iron. It is often, however, found in loose soils, in sands, and in river beds. Most of the gold recently obtained in California is from localities of the kind last mentioned. Pure gold is the most malleable of all metals, leaves having been beaten so thin, that 282,000 are required to be superimposed to make an inch. It melts at 2192° F., and is vaporized in the focus of a large burning-glass, or in the flame of the oxy-hydrogen blowpipe.
4. Native Mercury.
Mercury sometimes occurs native, although it is generally obtained from certain ores. It is the only metal which is liquid at ordinary temperatures, becoming solidified only at and under −39° F. The crystals which form in this state belong to the monometric system, and are principally octahedrons (fig. 33). It occurs as an amalgam, mixed with silver, in different proportions. The amalgam is generally stiff, rather brittle, and whitens copper when rubbed with it. Mercury mixed with silver presents the crystalline forms of both metals, which, at any rate, belong to the same system. The principal forms of such crystallization are shown in pl. 33, figs. 30, 45, 61, 62, 63. Mercury is obtained principally from the mines of Idria in Austria, and Almaden in Spain. Valuable deposits of cinnabar, or sulphuret of mercury, have recently been discovered in California.
5. Native Copper
Although most of the copper of commerce is obtained from its ores, yet it occasionally occurs in enormous masses, as a native metal. It is sometimes found crystallized, the crystals belonging to the regular system. The principal forms are represented in figs. 24, 30, 36, 40, 41. Most generally, however, the crystalline structure is not so evident, the metal occurring in various amorphous shapes; in veins (fig. 13) and beds in granite, syenite, grauwacke, slate, red sandstone, &c. Its principal localities are Hungary, Siberia, Brazil, China, Japan, and North America. The region about Lake Superior furnishes the largest masses of any known; one now in Washington, weighing 3704lbs., and others occurring of such enormous size as to present almost insuperable difficulties in the way of following out more manageable deposits. A mass was quarried out in the Cliff Mines (Lake Superior region), the weight of which was estimated at eighty tons.
6. Native Bismuth
This metal rarely occurs pure in nature, being generally combined with arsenic. It occurs in crystals of a rhombohedral form, nearly cubes in appearance. Metallic bismuth is of a whitish steel color, with a slight tinge of rose-red, and on being broken exhibits a coarsely foliated crystalline texture. This is shown in great perfection by melting the metal in a crucible, and after allowing a hard crust to form, piercing this, and permitting the still liquid portion to run out. Pl. 33, figs. 5 and 10, represent the crystallizations thus obtained. Native bismuth is found in veins, in gneiss, granite, rarely in clay-slate; generally accompanied by other metals. The richest localities are in Northern Europe. The only one known in the United States is in Connecticut. Bismuth is used principally in the formation of such alloys as require to become liquified at rather low temperatures.
7. Native Antimony
This rarely occurs native; when found it is of a tin-white color, rather brittle, and when crystallized, forms obtuse rhombohedrons (figs. 25, 31, 38). Specific gravity 6.5–6.8. It is not ductile, but flies to pieces under the hammer; is readily fused, and burns before the blowpipe with the evolution of a dense smoke of oxyde, deposited in crystals in the vicinity of the burning mass. It occurs in veins and lamellar concretions in crystalline rocks, associated with arsenic, silver, galena, &c., in the Hartz, Bohemia. Sweden, and in Dauphiny. The uses of antimony are various: it is employed in the arts in the composition of various alloys; thus antimony and lead constitute type-metal. Its pharmaceutical applications are numerous; thus the sulphuret is a very powerful medicine, known as kermes, and the oxyde of antimony is much used in various combinations.
8. Native Arsenic.
This metal is frequently found native. It occurs in acute rhombohedrons when crystallized (pl. 33, fig. 25), or in amorphous masses, or in small concave scales set one within the other. It tarnishes readily in the air, becoming nearly black, and losing its original tin-white color and metallic lustre. In its native state it is frequently called cobalt by the miners, on account of its deceiving them in their expectation of finding a lump of metal after roasting the ore containing it; the process actually converting the metal into arsenious acid, which passes off in vapor. The presence of this vapor may readily be known by its strong smell like garlic. This vapor is highly poisonous. The substance known in commerce as arsenic is not the metal, but an acid combination with oxygen forming arsenious acid. It is produced by roasting the arseniurets of cobalt, iron, or nickel, and collecting the vapors in long chimneys or iron receivers. It occurs first as a hard, clear glass, which subsequently becomes an enamel white. Although exceedingly poisonous, it is an indispensable agent in the arts, especially in those of coloring and glass-making. It is an invaluable agent in the preservation of objects of natural history, being applied, mixed with water, alcohol, or whiskey, to those surfaces which it is desired to preserve from putrefaction, or the attacks of various insects. It is also used in poisoning vermin: fly-powder is a preparation of arsenious acid. Sulphurets of arsenic occur both native and manufactured: one of them is the golden-yellow orpiment; the other, realgar, of a fiery red. The oxygen combinations of arsenic are all acids, and, with the sulphurets. are of great importance as medicinal agents.
Class 3. Tellurids.—Hausmann
The minerals of this class are combinations of electro-positive metals with tellurium. These tellurids are known by their partial volatilization when heated in a glass tube, giving off vapor of tellurous acid, which is deposited on the sides of the tube. On applying additional heat to this deposit, it first melts and then evaporates.
These combinations are very rare, and not very widely diffused. Those best known are graphic tellurium, white tellurium, laminated tellurium, and auro-tellurite, being mostly combinations with gold, silver, lead, copper, sulphur, &c. Those which principally occur in Transylvania are valuable mainly for the gold they contain.
Class 4. Antimonids.—Hausmann
These also are known by their behavior when heated in a glass tube. The vapor, however, which is deposited in a crust on the sides of the tube, volatilizes on being heated without first melting like the tellurids.
1. Antimonial Silver
This ranks among the rich ores, as it contains 77–84 parts of silver to 23–26 of antimony in 100. Its specific gravity is 9.4–9.8; it is harder than calcareous spar, and is of a silvery tin- white color. When heated on charcoal it becomes converted into a brittle granule of antimonial silver, and, on continuing the heat, the antimony is all driven off in a white vapor, leaving the silver pure. It is somewhat brittle, a little ductile, and crystallizes in forms of the trimetric system (pl. 33, fig. 39), which are often grouped as in fig. 47. It is found in granular masses at Wolfach and Andreasberg.
2. Antimonial Nickel
This consists of two equivalents of antimony and one of nickel. Its color is a light copper-red, running into violet; its fracture is uneven, small subconchoidal; it is harder than fluor-spar; it crystallizes in double six-sided pyramids (pl. 32, fig. 72), and in thin hexagonal plates. The principal locality is at Andreasberg in the Hartz.
Class 5. Arsenids.—Hausmann
Combinations of arsenic with electro-positive metals. They are characterized, like the antimonids and tellurids, by giving out a white coating to the upper part of the glass tube within which they are heated, which is volatilized by additional heat, without melting. They give out, however, when heated on charcoal before the blowpipe, the peculiar garlic-like odor of arsenic.
1. Copper Nickel
This is one of the most important of these combinations. It contains 34–40 per cent, of nickel, and may be used as an ore of this latter metal. It is of a bright copper-red color, to which it owes its name, and besides arsenic, also contains cobalt, iron, lead, antimony, and sulphur. It is found in Saxony, Bohemia, Thuringia, Styria, Hungary, &c., associated with cobalt, silver, and copper.
2. White Nickel
This, like the last, affords a workable proportion of nickel, containing 20–30 per cent, of this metal, with 71–73 of arsenic, and small quantities of cobalt, iron, copper, bismuth, and sulphur. It crystallizes mostly in cubes, with octahedral and rhombic-dodecahedral faces (pl. 33, figs. 36, 44, and 61). More rarely it is found in amorphous finely-granular masses. The ores of nickel have only recently become of importance, although the metal itself was discovered by Cronstedt in 1751. Its peculiarities, which assimilate it to the noble metals, have caused it in latter times to be much employed for various purposes. The metallic nickel is generally obtained from copper nickel, a name given this ore on account of its color, and for a long time considered an opprobrious epithet as long as it was found impossible to reduce it.
Nickel occurs almost always associated with cobalt, and both metals with arsenic, so that most cobalt ores are worked at the same time as well for their cobalt as the nickel which they contain. Nickel is of a silvery-white color, passing into steel grey; is very difficult of fusion, and heavier than iron. It is very malleable, and may be hammered into plates of one hundredth of an inch thick, and drawn into wire of one fifty-sixth of an inch in diameter. It may also be welded. It combines with several metals at a strong heat, and these alloys are employed practically in a great many ways. The most important of the combinations are known as white copper, German silver, argentine, packfong, and maillechort. The German silver used in the construction of knives and forks, consists of two parts of copper, one of nickel, and one of zinc. A mixture of five parts copper, two of nickel, and two of zinc, has nearly the color of silver, and is used for making knife handles, snuffers, gun mountings, philosophical apparatus, &c. For cutting out fine sheets, a mixture of three parts copper, one of nickel, and one of zinc, is best, as being most malleable. For castings, from two to three per cent, of lead must be added; and to obtain the most silvery-white color possible, the three metals must be melted together under a layer of charcoal powder, and a small quantity of iron added. This iron alloy is, however, much more difficult to work, on account of its brittleness. Care must be taken in using culinary and table articles constructed of German silver, that they be brought into contact with no corrosive liquids, as in addition to the poisonous effects of the essential metals, there is frequently a small proportion of arsenic still remaining in the nickel.
3. White Cobalt or Smaltine
This mineral is of importance as an ore of metallic cobalt. It occurs in veins of primitive and transition rocks, associated with various other metals, as nickel, copper, iron, silver, and arsenic; in Saxony, Thuringia, Alsace, Spain, Hungary, &c. It is found in crystals belonging to the regular system, as cubes (pl. 33, fig. 35), rhombic dodecahedrons (fig. 30), and the combinations of these. It is also obtained in reticulated, ramified, and various other irregular and derivative shapes. It contains about twenty per cent, of cobalt, and seventy-four of arsenic. Metallic cobalt, principally obtained from this arseniate of cobalt, is a refractory metal, similar to iron, and like it magnetic. The combinations of its oxydes form very valuable coloring matters, particularly for blue. To prepare these the ore must be freed from the arsenic, forming its larger portion, by roasting it in the air, the result of which is to drive off the arsenic in white fumes of arsenious acid. The resulting ore may be applied to many purposes. Thus it imparts a deep and beautiful blue to glass, for which it is much used. Melted with quartz it forms zaffre, safflor, or zaffra, used in the preparation of smalts. This is a glass formed from quartz and potash, colored by zaffre. The cobalt colors, known as Leitner’s Blue and Thenard’s Blue, are of great beauty. The first is obtained by calcining alumina with nitrate of cobalt; the latter by calcining alumina with phosphate of cobalt. Both remain unchanged in fire, for which reason they are applicable in painting porcelain and glass.
4. Glance Cobalt
This mineral, like the last, is very rich in cobalt, for which reason it is much used in smalts works. It occurs in beautiful crystals of the regular system. The most common forms are shown in pl. 33, figs. 57, 61, 62, 50, 64, 60, 54, 55, 49, 58. It possesses a metallic lustre, is opake, with the fresh fracture of a silvery-white color, inclining to red, which soon overspreads the surface, and presents a beautiful play of metallic colors. It is brittle, and the fracture is uneven, somewhat conchoidal; when heated it gives off arsenious and sulphurous acid. It consists of 33.10 per cent, of cobalt, 43.46 of arsenic, 20.08 of sulphur, and 3.23 of iron. It occurs in veins and beds of primitive rocks in Norway, Sweden, and Alsace.
Class 6. Selenids
The minerals of this class are combinations of selenium with electropositive metals. They are readily recognised by the peculiar odor of burning selenium, when the substance to be tested is heated on charcoal before the blowpipe. A smell is developed, if selenium be present, similar to that of decaying horse-radish.
Selenium, when solid, is of a dark-brownish red color on the surface, the conchoidal fracture exhibiting a metallic leaden-grey tint. When in the finely divided state, obtained when reduced from the watery solution of selenious acid, it is of a vermilion red color. Thin splinters of compact selenium are somewhat transparent. It burns like sulphur, when heated, diffusing its characteristic odor, sensible even in very small quantity. In its general peculiarities and combinations it has much resemblance to sulphur. In and of itself it is of no practical importance; its combinations are some of them valuable for the metals they contain. The principal of these combinations are:
1. Selenid of Silver
This mineral occurs in Mexico, and more rarely at Tilkerode in the Hartz. It forms thin plates, which are rather pliant, and exhibits a cleavage into cubes. It is harder than rock salt, is opake, lustrous, iron-black, and contains from sixty-five to sixty-seven per cent, of silver.
2. Selenid of Mercury and Lead
This remarkable mineral is exceedingly rare, and is found at Tilkerode and Lerbach in the Hartz. It occurs in finely granular, metalloid, bluish-grey streaks and spots, in a ferruginous calc spar, as shown by the dark portion m pl. 33, fig. 21. A third selenid, that of lead, occurs at Tilkerode, under similar circumstances. It is difficult to distinguish from the preceding, and furnishes a very argentiferous lead after reduction. It is, however, very rare.
Class 7. Sulphurids, or Sulphurets
These are combinations of electro-positive metals with sulphur. They all yield the smell of sulphurous acid when heated before the blowpipe on charcoal; they are soluble in nitro-hydrochloric acid, with the separation of sulphur.
1. Galena
Galena or sulphuret of lead is one of the most important of all minerals, furnishing the greater part of the lead of commerce, as also a considerable amount of silver. It occurs principally in veins, as in the Hartz, in Saxony, in Bohemia, France, England, and Spain. The most abundant deposits known are those in the north-western parts of the United States, Missouri, Iowa, Wisconsin, and Illinois. Galena is a combination of equal atomic weights of sulphur and lead, or of 104 equivalents of lead and 167 of sulphur, with a slight admixture of other sulphurets, as of silver, antimony, bismuth, copper, and iron. It most generally occurs in amorphous masses, which exhibit crystals in the cavities. The crystals belong to the monometric or regular system, the most usual forms being those of pl. 33, figs. 35, 33, 41, 43, 44; which, however, like most natural crystals, seldom or never occur in so perfect a form. It is more generally the case, that apart from incomplete development, the same crystal presents faces belonging to several different forms. The cube often appears incomplete or distorted, as shown in fig. 20. Other natural crystallizations are shown in figs. 11, 17, and 19. The crystals of galena, as well as the amorphous masses, tarnish on exposure to the air, exhibiting a dark bluish-grey hue; the fresh fracture is leaden-grey, and very lustrous. They have a three-fold cleavage, a piece of galena, when broken by a hammer, exhibiting the cubic character, as shown in fig. 6.
The specific gravity of galena is 7.58, its hardness being a little greater than that of rock salt. When heated, it breaks up, if struck with a hammer, into little cubes; at a high heat it melts and becomes converted into vapor, which is decomposed in the open air, with the formation of sulphate of lead; if this operation be conducted in closed vessels, the vapor condenses in crystals on the sides or colder parts of the furnace. These crystals present the appearance of imperfect cubes, as in fig. 8.
To obtain lead from its sulphuret, the latter is roasted in the open air, by which a great part of the sulphur is burned. The remainder, consisting of partially desulphuretted lead and sulphate of iron, is melted with charcoal and limestone. The lead thus obtained still contains the other metals which were originally mixed with it. It is called workable lead when in this state, and is, first of all, to be treated for silver. For this purpose it is brought into a cupellation furnace, so constructed, that a constant stream of air may pass over the melted metal. By this means the oxygen of the atmosphere coming in contact with it, converts it into oxyde of lead, which exhales to a considerable extent, but is principally drawn and let out through an adjoining aperture. The silver does not combine with the oxygen during this operation, and while the oxyde of lead or litharge is constantly being removed, the silver remains behind, uptil finally every particle of oxyde of lead has disappeared. The silver then undergoes a further preparation after being removed. The oxyde of lead is either sold in this form, as litharge, or it is melted afresh with charcoal, and reduced again to the metallic state.
Lead, on account of its malleability and readiness of fusion, its ease of working, and its great abundance, is one of the most important of all metals. Its oxydes and salts are also of extensive application. Litharge is used in glazing pottery and in the manufacture of glass; an addition of minium or red oxyde of lead, to melted glass, renders it transparent, and constitutes what is known as flint-glass, in contradistinction to crown-glass, into whose r composition little or no lead enters. The glass used in porcelain painting, as also the pastes of the artificial gems, all contain a necessary proportion of oxyde of lead. Of the salts of lead, white lead is the most important. This is a carbonate of lead, or a carbonate combined with a hydrated oxyde. Acetate of lead, or sugar of lead, is used in dyeing, and in the preparation of many mineral colors.
2. Vitreous Silver, or Silver Glance
This is a simple sulphuret of silver, and consists of 87.032 per cent, of silver and 12.968 of sulphur. It crystallizes in many-faced crystals of the regular system (pl. 33, figs. 30, 33, 35, 36, 45), and also occurs in reticulated, arborescent, filiform, and amorphous masses, as also in plates. It readily melts before the blowpipe, emits the odor of sulphurous acid gas, and leaves behind a button of silver. It occurs in veins, accompanying other ores of silver, and is not abundant except in Saxony, Bohemia, the Tyrol, Hungary, Spain, and Mexico. It is very remarkable in being a malleable ore, flattening out under the stamper.
In places where sulphuret of silver, native silver, and other rich silver ores occur, the metal is obtained by amalgamation. This consists in reducing the ore containing silver into a very finely divided condition, and in this state shaking it up with mercury; the mercury combines with the silver, forming an amalgam. Should sulphuret of silver be in question, it will be necessary first to drive off the sulphur by roasting the pounded ore with salt. During this operation the silver is converted into chloride of silver and metallic silver, which mixture is to be treated with water and iron to remove the chlorine from the chloride, and to separate the silver in the metallic state. The residuum is then, as before remarked, to be agitated for a long time with mercury and iron in vessels, during which operation the amalgam is formed. After straining this amalgam, to separate the uncombined mercury, the residuum is placed in iron vessels and distilled. The mercury passes over in a sublimation, and the silver is left pure.
3. Sulphuret of Copper and Copper Pyrites
The expressions, glance and pyrites, generally indicate combinations of sulphur. The copper pyrites forms an important ore from which to obtain the pure metal. It is a combination of simple sulphuret of copper with simple sulphuret of iron, and contains from thirty-two to thirty-four per cent, of copper. It is extensively distributed in veins and beds in primitive and transition rocks, being generally accompanied by galena. Copper pyrites has a brassy-yellow color, rather deeper than that of sulphuret of iron, and generally occurs in coarse masses, whose cavities present crystals belonging to the dimetric system, and exhibiting the faces of an acute (pl. 33, fig. 37), or an oblong (fig. 48), square octahedron. When roasted in the air its sulphur is converted into sulphurous acid, while the iron and copper oxydize, and combining with the sulphuric acid, form a soluble salt obtained by leaching the roasted mass. This salt is much used in the arts, being known as blue vitriol, containing green vitriol. This same double salt forms in mines by the gradual oxydation of copper pyrites, a considerable amount of which is dissolved in the waters of such mines. The salt is obtained from this solution, either by evaporation or by precipitating metallic copper by means of iron. The principal portion of the pyrites procured in mines is, after roasting, reduced to metallic copper. It is first melted with its flint matrix, by which means the oxydized iron is principally combined with the quartz, and converted into a slag, under which lies the copper combined with sulphur. This, after repeated roasting, is again melted with quartz sand and charcoal, and still more freed from iron, until finally an impure, sulphurous, and little ductile copper is found under the slag, mixed with the accompanying metals, lead, iron, arsenic, antimony, zinc, &c. This impure copper is purified by long-continued fusion in the melting furnace, in which the foreign admixtures are partly oxydized, partly driven off. The metal still requires additional preparation by being melted with charcoal before it will become perfectly malleable.
4. Iron Pyrites
This is a sulphuret of iron which occurs extensively distributed, and in large quantity. It is found either crystallized or amorphous, in various rocks, clay slate, greenstone, hornblende, syenite, &c.; also in independent beds of considerable size, or in veins accompanying other ores. The crystallizations of pyrites are among the most perfect which occur in nature. Pl. 33, fig. 2, exhibits some single crystals from Chamouny: a is a cube distorted to a rectangular parallelopipedon; b is a pentagonal dodecahedron, whose pure crystalline shape would be that of fig. 57. Fig. 15 exhibits an agglomeration of cubes of pyrites lying between crystals of calcareous spar; and fig. 22, a lump of crystallization of iron pyrites, like the preceding, drawn from nature. The crystals of pyrites which here appear in the simplest shapes of the regular system, exhibit other more complicated forms, as seen in pl. 33, figs. 33, 35, 41, 50, 54, 57, 60. The crystals as well as the amorphous masses, have a light brassy-yellow color. A remarkable variety of iron pyrites, which is of a whitish yellow color, and more easily decomposed by the air than the preceding, is the water pyrites. It is not different from the other in chemical composition, but crystallized in another system, namely, the trimetric.
Iron pyrites is a very useful mineral, although not applicable as an iron ore. It is rather used to furnish sulphur, and, in many places, the gold which is combined with it in small quantities pays well for its extraction. The pyrites is roasted, and the iron burnt to an oxyde, the sulphur to sulphuric acid. On leaching the mass, sulphate of iron is obtained, a salt of extensive application in the arts, under the name of green vitriol, or copperas. From the roasted green vitriol, fuming sulphuric acid, or the Nordhausen acid, is obtained. The residuum of oxyde is known in commerce as colcothar, and is used for coloring or polishing. Iron pyrites strikes fire with steel; and has, in certain cases, replaced flint for this purpose.
5. Molybdenite, or Sulphuret of Molybdena
This is exhibited in foliated masses, composed of very thin, bluish grey, metalloid plates, unctuous to the touch like graphite. It occurs imbedded in granite and gneiss, in Sweden, Silesia, Hungary, France, Switzerland, the United States, and other places. It is found, generally, in the shape of bent leaves, between the constituents of granite, as shown in pl. 33, fig. 23. The metal forming the basis of this mineral has some very peculiar oxydes and salts, which are distinguished by their beautiful color, and are used by painters.
6. Arsenical Pyrites
This mineral is distinguished for its remarkable crystalline forms. It is of a silvery white color, uneven fracture, and also occurs amorphous, granular, or homogeneous. On being heated it gives off metallic arsenic, leaving behind simple sulphuret of iron; for this reason it is very useful for the production of arsenic. It sometimes contains small quantities of silver and gold. The crystals of this mineral are rectangular octahedrons, single individuals often occurring of great perfection sprinkled in the general mass, as shown in fig. 18. It is found in beds and veins, in the primitive rocks, at Freiberg, the Hartz, in Bohemia, Cornwall, Sweden, and Hungary.
7. Grey Copper Ore
Besides the preceding simple sulphurets, there are still a large number of more complicated combinations; among which this ore, or fahlerz, is conspicuous. This may be considered as a sulphuret of antimony, copper, and iron; in which combination, however, the antimony is sometimes partially or entirely replaced by arsenic, the iron by zinc, and in another variety by mercury: in the argentiferous ore, for the iron and copper, silver is partly substituted. This ore is sometimes found in remarkable hemihedral crystallizations of the regular system. Thus there are tetrahedrons variously modified by faces of other forms of the same system. The most common forms of these crystals are seen in figs. 51, 52, 53, 56, 59. The tetrahedrons are often coated with a thin layer of copper pyrites, and then exhibit a beautiful golden lustre, these occurring at times of large size. The argentiferous grey copper ores are very valuable silver ores; that known as the graugültigerz contains 13–18 per cent., and the weissgültigerz nearly 31 per cent., of silver. The proportion of copper in this ore sometimes amounts to 30–40 per cent.; in the hydrargyrous ores the per-centage of copper is 34–36, and of mercury 2–8.
8. Cinnabar
This splendid red mineral is a sulphuret of mercury, and is valuable as being the chief ore of mercury. It occurs crystallized, as in pl. 33, fig. 25, combined with carbon and alumina, as hepatic cinnabar or liver ore, or lamellar as in the coral ore. Its geological position is in grauwacke, and in transition sandstone and limestone; being found generally in beds, more rarely in veins. It is generally accompanied by calc-spar, native mercury, and amalgam. The mines of Almaden and Idria furnish the greatest, quantity of cinnabar. Large mines have recently been opened in California. To obtain metallic mercury, this ore is heated in iron vessels, with iron or lime, by which the mercury is separated from the sulphur. Another process is to heat the cinnabar in furnaces so constructed that all the vapors generated are carried through long galleries, into chambers where they are condensed. Mercury and its ores are not generally distributed, and for this reason the metal bears a high price, It is used as a medicine, and also for coatings of glass mirrors: for many philosophical instruments, and for chemical investigations it is indispensable.
9. Realgar and Orpiment
Both of these substances have already been mentioned under the head of arsenic as artificial products. The natural realgar occurs in earthy and amorphous masses, or in rhombic prisms (fig. 39). It is a bisulphuret of arsenic, and possesses an aurora-red color, with tolerable transparency. Native orpiment is a tersulphuret of arsenic, is of a fiery yellow color, and is used in painting and dyeing, as also as an addition to lead m the manufacture of shot.
Class 8. Oxydes

Combinations of oxygen with electro-positive metals.
1. Oxyde of Iron
Of all minerals embraced in the class of oxydes, that of iron is unquestionably the most important. It is diffused over the whole earth, and is the principal material from which metallic iron is procured. It occurs in various forms, and is a constituent in all rocks, and in most minerals. Oxyde of iron, in and for itself, and in combination with water, forms several distinct minerals. Thus, crystallized peroxyde of iron forms specular iron. This occurs in very perfect crystals of metallic lustre, and occasionally splendent. It is opake, black, and hyacinth-red, with slight transiucency in the laminæ. It is brittle, and its fracture is conchoidal. When pure, specular oxyde is an unmixed oxyde of iron, containing 69.34 per cent, of iron, and 70.66 of oxygen. Its crystals have an acute rhombohedron (pl. 34, fig. 30) as the primary shape, and occur generally with planes of derivative forms (figs. 23, 25, 31, and 40). Specular oxyde of iron occurs also in aggregations of highly lamellar irregularly curved leaves, of a shining black color, known as micaceous iron ore: pl. 34, fig. 1, represents a mineral coated with this micaceous ore.
A second form of oxyde of iron is more important on account of the greater masses in which it is found. This is the hematite ore. Red hematite occurs, like specular iron ore, in beds and veins in the older rocks, particularly in clay slate, mica slate, gneiss, and granite; also in transition rocks in clay slate, and in grauwacke. Hematite is of a steel-grey, cherry-red, brownish, and blood-red color, and occurs under the most diversified circumstances of distribution. The purer hematites are frequently of a highly fibrous texture, and reniform or kidney-shaped (fig. 20). It is sometimes found in cylindrical scaly masses, as in fig. 11, as also amorphous, dull, earthy, and, in the latter form, mixed with various rocks. With clay it forms red ochre, and with common limestone, the calcareous iron ore. Not less important are the brown and yellow iron ores. These are combinations of oxyde of iron with water, and furnish excellent ores for reduction. Fig. 9 exhibits a form of fibrous brown hematite, such as is frequently found in the Hartz, and fig. 5, another variety of its occurrence. The iron ores among which the oxydes are pre-eminent, always contain a considerable proportion of their gangue or matrix. The manufacture of iron from its ore consists of two distinct operations, the mechanical separation of extraneous matters, as far as possible, and the reduction of the oxyde to the metallic state. The iron ore is introduced in alternate layers with coal into a furnace several stories high, into the lower part of which a powerful blast is continually kept up by a blowing apparatus. By this means the contents of the furnace are brought to, and kept at, a white heat. At this temperature the oxygen of the iron combines with the carbon of the coal, and forms carbonic oxyde and acid, the iron being left in a metallic state. The accompanying rock is melted by adding the necessary flux. In this way the whole contents of the furnace become perfectly liquid, the iron, from its greater density, occupying the lower part. By making certain apertures in the clay stopping up the bottom of the furnace, either the slag or the melted iron may be drawn off.
The iron thus obtained is still very impure, and contains especially a good deal of carbon in the state of carburet of iron; it is now known as cast iron or pig iron. This pig metal is purified by remelting in a melting, or run out furnace; after which it is puddled, and hammered out into bars in forges. Bar iron still contains carbon, the amount not over one half per cent., and not materially affecting its malleable properties. On the other hand, a slight per-centage of phosphorus (also of silicon, according to some authorities) renders the iron cold short; and the presence of sulphur, arsenic, and copper, renders it red short. Iron, of all ductile metals, is the hardest and toughest; it cannot be hammered out into very thin plates without breaking, but on the other hand it may be drawn out into very thin wire. Purified and melted iron has a specific gravity of 7.79, common bar iron of 7.788. It melts at from 2822 to 2876° F.; and volatilizes in the flame of the oxy- hydrogen blowpipe, and in the heat produced by the galvanic battery. Steel, one of the principal products of iron, consists of a similar, but more intimate, combination of iron with carbon. The amount of carbon in steel varies according to the kind from 0.5 to 1.5 per cent.; and iron with this per centage of carbon acquires the property of hardening when rapidly cooled.
2. Silica
Silica or silicic acid, since it possesses the distinguishing properties of an acid, is a combination of oxygen with a simple substance, silicon, only obtainable by the delicate manipulation of the chemist. It is diffused in vast quantity over the earth. It occurs, in a very pure state, both crystalline and amorphous, and forms combinations with all fixed oxydes. The purest crystalline silex or silica is found in rock crystals. These occur in perfect single crystals of the hexagonal system, as double six-sided pyramids (pl. 34, fig. 36), or as regular hexagonal prisms with six-sided pyramidal terminations (fig. 51), or as derivatives from these forms, fs. 21, 22, 45, 72. Rock crystal frequently occurs in small crystals, more rarely several feet in length. Quartz, a common form of crystallized silex, and often somewhat impure, is well known to all miners; and in the most beautiful specimens presents somewhat the appearance of figs. 2 and 3. Rock crystal and quartz pass from perfect transparency into an opake milky white, and with foreign admixtures into every color of the rainbow. Besides the crystallized varieties of silica, there are others, as chalcedony, remarkable for their beauty and hardness, and indispensable in the manufacture of various instruments and utensils. The crystallized varieties are used as gems, and excellent lenses are obtained from perfectly transparent and colorless rock crystal. Quartz, either compact, or as sand, is a principal ingredient in glass; chalcedony and agate are converted into ornaments, polishing instruments, scale beds, mortars, &c. In sand we find one of the most important constituents of the fruitful soils; and finally all building materials, whether stone or brick, owe their solidity to the mortar of which this same sand is a most important ingredient.
3. Tin Ore
Tin ore is the oxyde of tin, and at the same time the source from which most of the metallic tin of commerce is derived. It occurs in primitive rocks, in veins or beds; traversing granite, gneiss, mica slate, and clay slate. Tin is not very generally diffused, and occurs in large quantity in but few localities, especially in Bohemia, Saxony, Cornwall in England, and in the East Indies, in Malacca, and Banca. The ore is sometimes found in remarkable crystals of the dimetric system; it is semi-transparent or opake, and of various colors, as white, grey, red, brown, or black. Pl. 34, fig. 8, represents a group of crystals drawn from nature, and fig. 19 a mass of granite imbedding similar crystals. The forms here represented are modifications and twin formations of the forms, figs. 29, 37, 42. This substance is a very valuable ore of tin, containing as much as 78 per cent, of the pure metal. In reducing the ore it is pounded fine, and roasted. By this means, a portion of its impurities is dissipated, especially arsenic and sulphur; others being converted into lighter substances which can be separated from the heavier tin by washing. The residuum is then to be heated with charcoal in the furnace, in the usual manner. The raw tin, thus obtained, is very impure, containing in addition lead, bismuth, arsenic, iron, antimony, and zinc. It is purified by the application of a gentle heat, during which the purer tin runs off, leaving a portion behind mixed with impurities. The best and purest tin is the Malacca, Banca, and English grain tin. After this comes the common English grain tin, then the English block tin, and the Bohemian and Saxony mountain tin. The applications of tin are very numerous. It is cast into various shapes, converted into different culinary utensils; covering sheets of iron plate it forms the material used by the tinner; mixed with mercury it constitutes the amalgam coating the backs of looking-glasses, &c.
4. Rutile and Anatase
Both minerals contain the same oxyde, resembling the oxyde of tin in many respects. The metal obtained from them, titanium, is the only one of a red color besides copper. Neither mineral is abundant, the principal localities being Norway, Spain, France, Hungary, and Switzerland, where they occur in primitive rocks. Several localities in the United States are known where rutile is found. Pl. 34, fig. 10, represents a group of rutile crystals drawn from nature, while figs. 28, 43, 53, give its theoretical forms; figs. 29 and 32 represent the crystalline form of anatase. The oxyde forming these minerals is an acid of titanium, called titanic acid.
5. Pyrolusite, Braunite, Manganite, Hausmannite
These are all oxydes of a metal, manganese, similar in its properties to iron. Hausmannite is a red oxyde of manganese, and occurs crystallized, in shapes of figs. 29 and 32; pyrolusite is oxyde of manganese, its crystal forms as in figs. 26, 28, 29; manganite is hydrated sesquioxyde of manganese, and crystallizes in shapes whose primary form is a rhombic prism (fig. 24); braunite is anhydrous sesquioxyde of manganese, crystallized like manganite, and accompanying it. Crystals of both occur together, as at Ilefeld, in beautiful masses, as shown from nature in fig. 7. These oxydes also occur amorphous, as coatings of other minerals. Pyrolusite and manganite are used in the manufacture of oxygen, which may be procured by simple heating of these minerals; their further applications in the arts are numerous. They are used in glass works to decolorize glass, the bottle-green color of common glass disappearing entirely by the addition of pyrolusite; an increased proportion of the mineral produces a beautiful red in melted glass, which, by the addition of oxyde of tin, becomes converted into a flesh-color, used in the painting of porcelain and glass. The metal manganese has been converted to no useful purpose, owing to the difficulty of separating it from its ores.
6. Sapphire, Ruby, and Corundum
These beautiful gems are oxydes of aluminium, a silvery-white metal, and in chemical composition do not differ from the purest common clay, being rather its crystallization, modified by slight admixtures. The forms of the crystals are those of pl. 34, figs. 30, 36, 38. The most transparent blue crystallized variety is called sapphire, the yellow crystals are called oriental topaz, and the transparent red variety, oriental ruby. The less transparent kinds, of different colors, of green, blue, red, and greyish, are all grouped together under the general name of corundum. The transparent varieties occur in single, frequently rounded, crystals and grains, in river beds, and in alluvial soils, especially in Ceylon, China, France, and Bohemia. Corundum is found in primary formations in Switzerland, North America, Sweden, the East Indies, and China. Red corundum is the ruby, which is often more highly valued than the diamond. Sapphires are very costly gems, and are generally cut like the diamond. Rubies of eight grains are worth from $200 to $250; the most esteemed sapphires of a dark-blue and pure color, are worth from $350 to $400 for a weight of twenty-four grains; the paler, of sixteen grains’ weight, $25 to $30. The sapphire, ruby, and corundum, are the hardest stones after the diamond.
7. Arsenious Acid
This has already been referred to under the head of arsenic. The most common forms of the crystals are the tetrahedron (fig. 39) and the octahedron (fig. 40). It frequently accompanies rocks containing arsenic. It is nearly allied to the following minerals:
8. White Antimony
This is an oxyde of antimony, corresponding to the last mineral, and occurs in crystals, whose form is represented in pl. 34, fig. 50. It accompanies the ores of antimony, and especially grey antimony.
9. Red Copper Ore
This is an oxyde of copper occurring in beautiful crystallizations of the regular system, as in figs. 27, 33, 43, 44, 61, 64. It is of a cochineal red color, passing into bluish grey, possesses an uneven conchoidal fracture, and an adamantine lustre on a very smooth surface. The finest crystals have been found in Cornwall, in Siberia, and at Chessy near Lyons, in primitive and transition rocks.
Class 9. Silicates
Besides the simple oxydes just mentioned, there occur in nature a large number of their combinations, amongst which, those of different oxydes with silica (the silicates) are by far the most numerous. They are known before the blowpipe by their decomposition by the salt of phosphorus, a useful test, the silica being set free, and the base uniting with the free acid of the salt of phosphorus. The variety of silicates is very great, as will subsequently be shown.
1. Datholite
Datholite is a combination of silicate of lime with borate of lime and water, or a boro-silicate of lime. It is eminent for the beauty and perfection of its crystals. It is found in great abundance at Andreasberg, in compact greenstone, traversed by veins of compact dolerite, and having crystals of the mineral in its cavities; also in New Jersey, Connecticut, and the Michigan copper region. It crystallizes in the monoclinic system, its forms being the oblique rhombic prism (pl. 34, fig. 73) and its derivatives. Fig. 6 is a group of datholite crystals from nature. This is a rare mineral, and of no practical application.
2. Apophyllite
Apophyllite is one of the most beautiful minerals in nature. It occurs in veins and beds in primitive and amygdaloidal rocks in the Tyrol, Bohemia, the Hartz, Scotland, Greenland, Mexico, and North America. Its most usual crystal forms are represented in figs. 28, 37, 43. It sometimes occurs in crystals of various sizes, grouped together (fig. 17), the individuals forming acute square octahedrons. It is transparent, colorless; or else, white, yellow, green, and rarely rose-red. The crystals have a very perfect cleavage in the direction of the basal plane, from which the mass may be diminished by sections, perpendicular to the main axis, producing square faces. The basal corners are almost always replaced by rhombic faces, striated in the direction of the primary axis. Apophyllite is silicate of alumina combined with silicate of potash.
3. Olivine, or Chrysolite.
This mineral is a silicate of magnesia with a slight admixture of protoxyde of iron, and rarely oxydes of manganese, chromium, nickel, copper, and tin. Olivine is widely diffused in small crystalline granules, and occurs in the cavities of most basalt. The larger crystals (or the noble olivine) are found in Bohemia and in Hungary, and are used for ornamental purposes. Chrysolite occurs of a green, brown, or red color. The crystals belong to the trimetric system, as in pl. 34, fig. 74.
4. Picrosmine
This is a hydrous bisilicate of magnesia with a slight admixture of protoxyde of iron, and manganese, with a small amount of alumina; it occurs in a single deposit in Bohemia. It is opake, sub-transparent at the edges, iridescent, and of a vitreous lustre, and of various shades of green. From its cleavage, its crystallizations belong to the form of fig. 41.
5. Chondrotite
This mineral, a silicate of magnesia, is found in New Jersey and elsewhere in the United States, in Finland, &c. It is transparent or translucent, of yellow, brown, reddish-yellow, green, or black color; and generally occurs in granules. When heated it loses color, becoming, first black, and then white. Its crystallization is that of pl. 34, fig. 54, or monoclinic.
6. Augite, or Pyroxene
This occurs finely crystallized in volcanic mountains, and in lavas, and forms an essential constituent of basalt and dolorite. It is dark green, brown, and black, sometimes light green and white, with a vitreous or resinous lustre, opake, or only translucent at the edges. The crystals belong to the monoclinic system, and present principally the forms of pl. 34, figs. 35, 52, 67. Augite, which is a silicate of lime and magnesia, contains often likewise alumina, protoxyde of iron, and protoxyde of manganese.
7. Hornblende
Hornblende is widely distributed in certain rocks, which it characterizes, and in an imperfectly crystallized state is fibrous and somewhat radiated. Like the preceding, it belongs to the monoclinic system, and fig. 75 represents its primary form. It differs from the preceding in containing less lime, but more magnesia and silica, being, like it, colored by the protoxydes of iron and manganese; it sometimes, in addition, contains alumina, fluor, and water. Hornblende occurs of various colors, grey, white, greenish, dark green to black, arid also in various modifications. A variety of hornblende, consisting of very long, closely compacted, and interlaced crystals, frequently occurs in gneiss, mica slate, and limestone. Fig. 13 represents this variety of gneiss.
8. Staurotide
Alumina combined with silex forms an extensive series of minerals, and in this species is frequently replaced, to a greater or less extent, by other minerals. In staurotide, for instance, a portion of the alumina is constantly replaced by protoxyde of iron. It occurs imbedded in gneiss and mica slate; the crystals belonging to the trimetric system (pl. 34, figs. 47, 70).
9. Andalusite (disilicate of alumina)
This mineral affords fine crystallizations of the trimetric system. Fig. 12 represents a mass of crystals of andalusite in granite. The crystals are right rhombic prisms.
10. Kyanite (disilicate of alumina)
The chemical composition of this mineral approximates it to the last; its crystals, however, belong to the triclinic system (pl. 34, fig. 65). Kyanite is often of a vitreous lustre, on the sometimes striated surface; the fracture is iridescent; the color sky blue, sometimes white. It occurs usually in bladed crystallizations, in mica slate, gneiss, dolorite, and limestone; it is rarely found amorphous.
11. Topaz (fluo-silicate of alumina)
Topaz is distinguished for its hardness. It exceeds quartz in this respect, and occurs imbedded, in very perfect crystals of the trimetric system, in gneiss and granite. It is found in great beauty and large quantity, in the topaz rock of Schneckenstein, in Saxony. The crystals from this locality have generally the appearance presented in pl. 35, fig. 9. Its composition may be set down at two thirds silicate of alumina, with fluo-silicic acid.
12. Humboldtite (silicate of alumina and lime)
This mineral is found on Mount Vesuvius. It crystallizes in shapes of the square system. It is very hard and scratches glass, has a yellow color varying to green, and melts before the blowpipe into a vesicular sub-transparent glass. Its crystalline form is shown in pl. 34, fig. 55.
13. Mica (compound silicate of alumina)
The common or bi-axial mica crystallizes according to the monoclinic system. A second variety, which is distinguished for its optical properties, and which crystallizes according to the hexagonal system (fig. 69), is the uni-axial mica. The common bi-axial mica is a very useful mineral. Its crystals are nearly regular rhombic plates, belonging to an oblique rhombic prism as the primary form. It exhibits the most perfect cleavage of all minerals, being capable of splitting into exceedingly thin leaves. It is used for window panes, and covers for microscopic objects, and is very useful in the cultivation of microscopic water plants. It occurs, in large leaves, in Siberia, and also in New Hampshire; and in general forms a constituent of all granite, embracing in its composition, silica, alumina, potassa, manganese, and fluoric acid.
14. Euclase (compound silicate of alumina)
This exceedingly rare mineral occurs in Brazil, crystallized according to the monoclinic system (fig. 58). It is transparent of a vitreous lustre, mountain green, passing into white and blue; brittle.
15. Idocrase (compound silicate of alumina)
This mineral occurs in very perfect crystals of the dimetric system (figs. 53, 48), with a vitreous or resinous lustre, sometimes striated on the surface. It is semi-transparent, brown, or of various shades of green. It consists of silica, alumina, oxydes of manganese and of iron, and lime. It was first found in the ancient lavas of Vesuvius; more recently, in other places.
16. Garnet (compound silicate of alumina)
Garnet, of which there are many varieties, presents itself in very perfect crystallizations of the regular system. The form shown in pl. 34, figs. 33 and 64, as also the rhombic dodecahedron (fig. 61), are especially prevalent. The faces of the crystals possess a vitreous lustre, are pearly smooth, or partly striated or rough. The colors are exceedingly various, being red, ferruginous, brown, yellow, black, white, and green (pl. 35, fig. 6). The transparency is either perfect, imperfect, or wanting entirely. The most remarkable varieties are the noble, the Bohemian, and the oriental garnet or almandine, of a beautiful cherry-red, blood-red, or brownish-red color, occurring in crystalline rocks, in gneiss, and chlorite slate. Another kind much esteemed is the pyrope, of a blood-red color, occurring in loose grains in clay near Bilin in Bohemia. Other varieties are colophonite, grossular, &c.
17. Beryl (silicate of alumina and glucina)
The noble beryl, or emerald, is a bluish-green or green gem. The most valuable crystals are found in Peru. Those of four grains are worth from twenty to twenty-five dollars; of twenty-four grains, from five hundred and fifty to six hundred dollars. The crystallized forms belong to the hexagonal system. The most frequent of these are shown in pl. 34, figs. 22, 49, 57, 67, 68, 69. A less important variety is found in beryls of a green, blue, and yellow color, either transparent or opake. Opake beryls are not rare, and occur sometimes very perfect in granite, as shown in pl. 35, fig. 15, a sketch drawn from nature. Beryls of great beauty are found in the granite of Mertschinsk, as also in the Ural and Altai mountains, and near Rio Janeiro. Beryls of enormous size occur in the United States. One specimen from New Hampshire weighs 240lbs., and is four feet in length. The lateral faces of well-formed crystals of beryl are generally striated vertically. In its composition it consists of a combination of silicate of glucina with silicate of alumina.
18. Prehnite (silicate of alumina and lime)
This mineral occurs in volcanic and primary rocks in the Tyrol, Piedmont, Carinthia, Salzburg, Sweden, Norway, and the United States. Its crystals belong to the trimetric system, and are derivatives of the forms in fig. 62, pl. 34. Prehnite generally occurs in rhombic prisms; it is also found granular, foliated columnar, and amorphous. It is a combination of silicate of alumina with silicate of lime, and water.
19. Nepheline
Nepheline, a variety of which is called elaeolite, occurs both amorphous and crystallized: its crystals belong to the hexagonal system. It is transparent or translucent, of conchoidal to splintery fracture, generally of an oil-green color, but is sometimes blue, red, and brown. It consists of silicate of alumina, silicate of potash, and silicate of soda.
20. Scapolite and Wernerite
The first named mineral bodies, besides lime and alumina, contain a small quantity of potassa and soda, wanting in Wernerite. Both are colored by a slight admixture of oxydes, especially of iron. Both crystallize in the dimetric system. Scapolite occurs in forms like fig. 46; Wernerite like fig. 53, pl. 34. Scapolite occurs in the volcanic rocks of Vesuvius, and is abundant in some crystalline rocks, especially granular limestone, occurring in many places in the United States; Wernerite in Sweden, Norway, Finland, and various parts of the United States.
21. Iolite, or Dichroite
This mineral is remarkable for its color, which, in one position, appears indigo-blue, and at right angles to this, yellow or brown. It occurs amorphous and crystallized, the crystals belonging to the trimetric system (pl. 34, fig. 71). They are found imbedded in granite. It thus occurs in Sweden, Norway, Finland, and in the Northern United States. It is sometimes polished, and used as a gem.
22. Thomsonite
This mineral is met with in Scotland, both amorphous and crystallized, The crystals belong to the dimetric system (fig. 50), have a smooth surface, vitreous lustre, and white color. They are more or less translucent to transparent. This mineral consists of silicates of lime, soda, and alumina, with water.
23. Natrolite
Natrolite occurs both amorphous and crystallized, and sometimes forms a very beautiful mineral of a white, whitish-yellow, or reddish color. Its crystallizations are often globular forms covered with fine needle-shaped crystals (pl. 35, fig. 4). They belong to the trimetric system (pl. 34, fig. 59). Natrolite, in addition to soda, alumina, and silica, contains water, and a small quantity of oxyde of iron and lime. It occurs in the Tyrol and Rhineland, being not very rare, especially in basaltic and volcanic rocks.
24. Leucite (disilicate of potassa and alumina)
This mineral is known by the form of its crystals, the shape shown in fig. 64, deriving from it the name of leucitohedron. It occurs in volcanic masses, as in the neighborhood of Rome and Vesuvius. It is generally of a grey color, and its crystals frequently exhibit considerable size and perfection.
25. Analcime (disilicate of soda and alumina)
Analcime is a hydrated silicate of soda and alumina. Its crystals are shaped like those of the preceding, and occur generally in hemispherical white crystals, as in the Tyrol, or in small transparent crystals of one or two lines in diameter, as shown in fig. 15, from the clay slate of Andreasberg in the Hartz.
26. Chabazite (silicate of soda, lime, potassa, and alumina) pl. 34 fig. 18
Chabazite is a hydrated silicate whose crystals belong to the hexagonal system (figs. 65 and 66.) It is semi-transparent, or translucent, of conchoidal fracture, and brittle. Stilbite and heulandite are minerals allied to chabazite in chemical composition, consisting of lime, alumina, silica, and water. The crystals are highly pearly rectangular prisms, belonging to the trimetric system. Fig. 14, pl. 34, is taken from an Andreasberg specimen. Mesotype (fig. 16) and epistilbite (fig. 63) are allied minerals.
27. Tourmaline
Common tourmaline, as it occurs in granite in the Hartz, is represented in fig. 4, pl. 34. This mineral is of great interest, owing to the optical and electrical properties of many of its varieties. The primary form of the tourmaline is an obtuse rhombohedron (fig. 34), under modifications of which the tourmalines occur of considerable size. The constitution of tourmaline is frequently very complex; it generally contains soda, lime, manganese, iron, alumina, silica, and boracic acid.
28. Harmotome
This mineral exhibits very interesting forms of twin crystals. Pl. 35, fig. 5, illustrates a specimen from Andreasberg. The crystals are right rectangular prisms, terminated by square octahedron faces; of these crystals, four unite at right angles, forming a cross. These sometimes unite at angles of 90°. This rare mineral consists of silicates of alumina and baryta, with water, potash, and lime.
29. Albite (disilicate of soda and alumina)
Albite or soda feldspar forms a component of many rocks, occurring in Siberia, in very large crystals. Their primary form is an oblique rhomboidal prism (pl. 34, fig. 65). Albite is generally of a white or grey, yellow, and red color; lustre vitreous; iridescent on the cleavage surface; lateral faces frequently striated.
30. Feldspar
Of all silicates, feldspar is the most important, as well as most generally diffused. It forms a constituent of many rocks, and consists of silicates of potassa and alumina; it crystallizes in forms of the monoclinic system (figs. 75 and 76, or pl. 35, fig. 26). Complete crystals possess not unfrequently a twin structure, as shown in fig. 2, pl. 35. Feldspar is readily cleft in two directions, the surfaces thus exposed being lustrous. Its fracture is conchoidal, uneven; color white, to flesh color. The white, yellow, flesh colored, or ferruginous portions of granite, are feldspar, which, with quartz and mica, compose this extensively distributed and important rock. The feldspar of granite decomposes surely though slowly; and its potassa, combining with water and silica, becomes a principal source to plants of this essential inorganic constituent. The rest of the feldspar, richer in alumina, and poorer in silicic acid and potassa, is also an important item in the soil; alumina being essential in rendering a sandy soil fruitful. Kaolin is also decomposed feldspar, and is found abundantly in granite. Feldspar is closely allied to the preceding mineral, namely, albite, when a portion of its potassa is replaced by soda, as in pericline. Closely allied to feldspar are oligoclase and spodumene, the latter containing lithia in place of potassa. Pl. 35, fig. 1, represents a specimen in granite from the island Utö. Labradorite, in which lime and soda replace potassa, belongs also to the feldspars.
Class 10. Salts
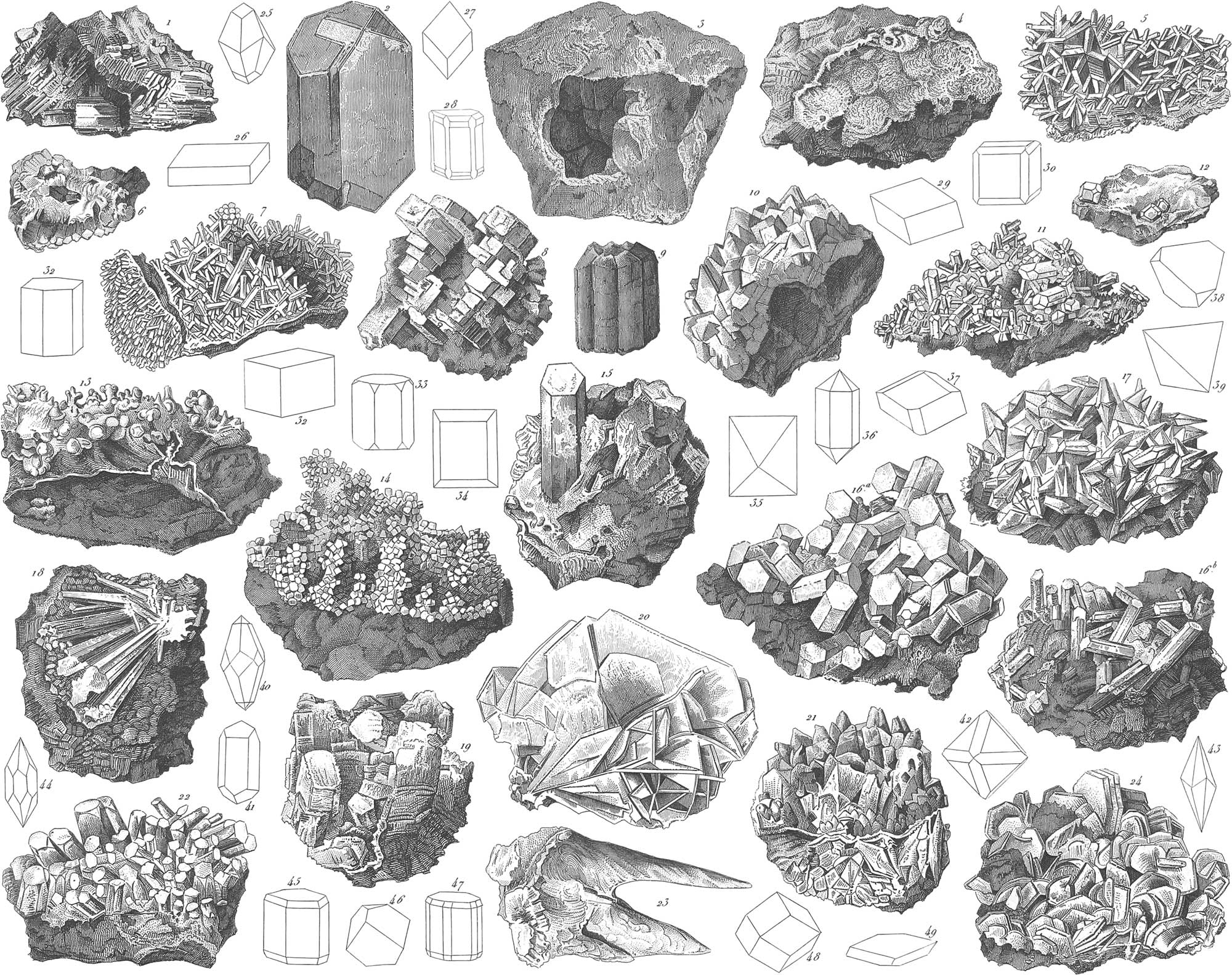

The minerals belonging to this class are non-silicious combinations of an acid with oxydes; from which an acid may be derived by boiling with caustic potassa, or by melting with carbonate of soda; or which exhibit an effervescence of carbonic acid gas when exposed to the action of certain acids.
1. Carbonate of Lime
Carbonate of lime is one of the most important of all minerals: it is a constituent of many rock-formations; is dissolved in all waters in the form of a bicarbonate; and as such, in combination with other acids found in the soil, enters as a necessary ingredient into the organism of plants and animals. Water containing carbonate of lime in solution is capable of dissolving fresh carbonate of lime, especially under pressure; such as would be produced on the lower end of a spring of water, in coming from a considerable depth below the surface. The pressure removed, and the water exposed to the concentrating influence of atmospheric evaporation, the necessary excess of lime will be deposited, in a crust, on all bodies wet by the fluid. Where, as in caverns, such water drops continually from a certain point, a deposit of carbonate of lime will be left behind, and will accumulate in time so as to form a considerable mass, resembling an icicle in shape. Such formations are called stalactites; and the less regular deposit of the same carbonate, which usually forms beneath the stalactite, is called stalagmite. Fig. 13, pl. 35, represents the initiatory stage in the formation of such stalactites, where the rock has become covered with a coating of stalactital matter, with depending projections. Fig. 23 represents a stalactite after it has attained its characteristic form.
Carbonate of lime is the actual constituent of limestone, chalk, and muschelkalk; it forms a principal portion of wood ashes, and the bones of animals, into which it is introduced by water holding it in solution. When crystallized it is known as calcareous spar. Calcareous spar has the most diversified crystallization to be met with in the mineral kingdom. It is likewise one of the dimorphous substances, or those whose forms do not all belong to the same system. Calcareous spar is consequently distinguished as arragonite, and as calcareous spar proper. In the former the crystals belong to the trimetric system (figs. 34 and 35). The crystals are right rhombic prisms, and are often grouped together, as in fig. 18. In the latter, or calc spar proper, the crystals belong to the hexagonal system. The forms are derived from the holohedral primary form of the double six-sided pyramid, or from a rhombohedron in which the angle is 105° 5′, some of which are represented in pl. 35, figs. 27, 29, 32, and 36. The hexagonal prisms are frequently pure, and such crystals often form very fine specimens, as in pl. 35, figs. 8, 16a, 16b; they are often terminated by the faces of the double six-sided pyramid, as shown by figs. 36, 45, and 47. Rhombohedral crystals, particularly secondary rhombohedrons, are also very common, together with various combinations of surfaces of rhombohedrons with those of six-sided prisms and pyramids. Fig. 20 represents a druse of rhombohedral segments (fig. 17) of dogtooth spar, with the very interesting crystal form of fig. 43, or the scalenohedron (scalene dodecahedron). The principal forms of crystallization of calc spar are shown in pl. 35, figs. 25, 28, 33, 37, 40, 41, 44, 45, 46, 47, 49, and pl. 36, fig. 37. In pl. 35, figs. 10, 11, 14, 21, 22, 24, and pl. 36, fig. 29, may be seen druses with many of the different modifications. Pl. 35, fig. 19, represents the hexagonal prism, with a finely crystalline coating of a second crystallization on the first, as a bed. The variety, pl. 36, fig. 3, is called double spar.
2. Carbonate of Iron
This mineral is often found associated with carbonate of lime, carbonate of magnesia, carbonate of manganese, and receives various names in its different modifications of constitution and form. The primary form of its crystallization is an obtuse rhombohedron: it also occurs in various secondary derivative forms. Pl. 35, fig. 3, represents a variety from Steinheim, called Sphærosiderite, a carbonate of iron of spheroidal form and radiated texture. Spathic iron is the usual name of the pure carbonate of iron; it often contains some carbonate of magnesia and lime. It occurs in vast beds, and yields an iron excellent for conversion into steel. Its crystals are shown in pl. 36, fig. 36, having the obtuse rhombohedron as their primary form. Spathic iron also occurs in crystal druses of the character seen in figs. 6 and 14. By increasing the proportion of lime and magnesia, to a degree sufficient to form dolomite, we have brown spar. The crystals of this have a very great similarity to those of simple calcareous spar; they are, however, readily recognisable, by becoming brown when exposed to the weather, owing to the manganese and iron they contain.
3. Carbonate of Strontia
This is met with in the form of strontianite, in no great abundance, and crystallized nearly like arragonite. The crystals occupy either the hollow of the druse (pl. 36, fig. 7), or present themselves in superficially radiating needles (fig. 20).
4. Carbonate of Lead.
The white lead of the arts is a carbonate of lead. This substance is found native, though rarely, in acicular, fibrous, distorted prisms, of the trimetric system (pl. 36, figs. 47 and 48); fig. 21 represents a druse of this white lead ore.
5. Boracite
This is a biborate of magnesia found at Luneburg and Segeberg, in very fine crystals, associated with anhydrite (pl. 35, fig. 12). The most usual forms of the Luneburg boracite are the rhombic dodecahedron (pl. 35, fig. 48), and their transitions into octahedrons (fig. 42) and rhombic dodecahedrons, with cubic and octahedral surfaces (fig. 30). The cube is also found with angles truncated or replaced (figs. 38, 39), but the opposite solid angles differ in their modifications. The crystals are imbedded in anhydrite.
6. Sulphate of Lime
This compound of sulphuric acid and lime constitutes, at times, whole beds of rocks, specially in connexion with rock salt. Without the water of crystallization, sulphate of lime is known as anhydrite or karstenite. In this form it occurs in amorphous masses (pl. 36, fig. 31), traversed or not by crystallized anhydrite, and is of a greyish-white, bluish, or blackish color. Combined with water of crystallization sulphate of lime forms gypsum or plaster of Paris. When gypsum is ground into an impalpable powder, and strongly heated in an iron vessel over a fire, its water of crystallization will be given off in the form of bubbles, rising through the powder, presenting a strong resemblance to ebullition. This, indeed, is technically termed “boiling.” This anhydrous powder, when mixed with water, will, by taking up a portion of the latter, soon harden, taking the shape of any form or mould into which the mixture may be poured. For this reason it has extensive applications in the arts. The unheated powder, if mixed up by means of a solution of carbonate of potassa, possesses the same property of fixing or hardening.
7. Sulphate of Baryta
The crystallized forms of this substance have the rectangular octahedron for their base. It received the name of barytes, or heavy spar, from its great specific gravity, by which it may readily be distinguished from all other minerals, excepting ores. The most common among the manifold crystallizations of barytes are shown in figs. 1, 2, 4, 6, 8, 9, 10, 13. It is generally of a white, or bluish-white color, and very abundant in some localities. It is used, when ground up, to adulterate white lead.
8. Vitriols
By the term vitriols miners understand the sulphates which are formed by the exposure of sulphurets to the weather, and become dissolved in the waters of the mines. Iron, or green vitriol (fig. 27), with its most common crystallized form (fig. 45), is sulphate of iron: it is of a green color, becoming ferruginous on exposure to the air from the formation of an oxyde. This substance is also known under the name of copperas. Copper or blue vitriol is also well known, this crystallizing in the form of figs. 34 and 35, pl. 36, and in crusts as in fig. 19. It is of a beautiful blue color, and, with the preceding, is much used in dyeing: white vitriol, the sulphate of zinc, is also used in the arts. It occurs in very decomposable prisms (fig. 23) of the trimetric system. The crystals are sometimes as in fig. 42.
9. Fluor-spar (fluoride of calcium)
This beautiful mineral is extensively distributed, and may be used for various purposes of ornament. From it is obtained fluoric acid, which may be used for etching glass. A druse of fluor is represented in pl. 36, fig. 32.
10. Wolfram (tungstate of iron and manganese)
This mineral is found in the gneiss of Bohemia and Saxony, in the form of large black striated crystals, with a lustrous surface (pl. 36, fig. 15).
11. Yellow lead ore (molybdate of lead)
This, associated with ores of lead and zinc, occurs, in beds and veins, in various parts of Germany, Hungary, Mexico, and North America. It crystallizes in variously modified square octahedrons, and occurs, as in fig. 22, finely crystallized, and in dirty yellow masses.
12. Pyromorphate, or green lead ore
This is a phosphate of lead containing some chloride of lead. It is found in Bohemia and Saxony, is of a pistachio-green color, and sometimes occurs in small green crystals of the hexagonal system, strewed as it were over the surfaces of other minerals. Fig. 10 represents a specimen from Zellerfeld.
13. Wavellite
This substance (shown in pl. 36, fig. 5) occurs in concentric radiated crystallized groups. It is a phosphate of alumina, and not very abundant.
14. Rock salt
Rock salt constitutes vast subterranean beds, which yield their contents either to actual mining operations, or by solution in salt springs. This salt or chloride of sodium crystallizes in cubes, which are generally distorted into rectangular parallelopipedons (fig. 30). In itself it is colorless, but often variously tinged by bitumen. Salt springs may be made to yield their solid contents by the evaporation of their waters. This evaporation may be brought about by boiling, or by exposure to the sun in shallow pans. In some places the stream of salt water is permitted to run for a long distance through a dense thorn hedge. In this way a considerable evaporation is effected by the extended surface exposed to the air. The water is also obtained in a purer form, as the other constituents of the spring, especially the gypsum, dissolved in all such waters, are, to a great extent, separated by the evaporation, and the thorns coated so as to present the appearance seen in pl. 36, fig. 11. The crystals in which the salt is separated from its solution by boiling are mostly incomplete cubes, generally with more or less perfect square funnel-shaped cavities.
15. Sulphates of the alkalies
The three most common alkalies, or their combinations with sulphur, form very important constituents of the soil. These sulphates are those of soda, of potassa, and of ammonia, all of which are well known as artificial products. The two latter have the same primary form of crystallization, namely, that of figs. 33 and 40, belonging to the trimetric system; sulphate of soda, on the other hand, crystallizes in the monoclinic system; pl. 36, fig. 18, represents crystals of sulphate of ammonia; fig. 28, of sulphate of potassa; and fig. 17, of sulphate of soda.
16. Alum
This well known substance, a sulphate of alumina and potassa, is produced by the decomposition of alum slate, and forms in minute crystals on the surface. By leaching earth containing alum, and evaporating the ley, the salt may be obtained in very large masses of crystals, whose primary form is the regular octahedron (fig. 24). Octahedral segments (fig. 25) in groups, as shown in fig. 16, are, however, more frequently met with.
17. Borax (borate of soda)
This substance is used as a flux in melting various metals and solders, and is applied to various other technical purposes. It is met with in a crude state as tincal, obtained in lumps from certain lakes in Thibet and Persia. The refined borax is obtained from tincal by repeated crystallization, producing variously modified rhombic prisms.
18. Saltpetres of potassa and soda
The ammonia which forms in the ground by the decomposition of various substances containing nitrogen, and diffused in damp air, becomes converted into nitric acid by contact with porous solid bodies, as limestone; the acid then combines with the bases existing in the soil. Thus the Chilian saltpetre is obtained by the saturation of this nitric acid by soda, this nitrate of soda crystallizing in obtuse rhombohedrons (fig. 36), and occurring in vast quantities, efflorescing from the surface of the ground, in South America. Common saltpetre arises in a similar manner by the conversion of the potassa of the soil into a nitrate. This does not occur naturally in large masses, but is prepared by artificial processes of decomposition. This saltpetre crystallizes in the trimetric system, with primary forms, as in figs. 47 and 48, as also in the modifications (figs. 41 and 43). Its crystals are generally hexagonal, with two acute terminations (fig. 26).
19. Phosphates of alkalies
These also are essential to the fertility of a productive soil, although occurring always in small quantities. Phosphate of potassa, as obtained by evaporation from its aqueous solution, crystallizes dimetrically (fig. 42): phosphate of soda, on the other hand, is monoclinic, and generally in modified shapes, the primary forms being represented in pl. 36, figs. 38, 39, 44, 46, and 49.
![]() Economical Uses of Minerals
Economical Uses of Minerals
In comparing the different groups of minerals just described, we find a great diversity in regard to their economical value. The utility of single minerals does not always depend on the properties which render a mineral substance fit for a certain end, but rather is based on the degree to which it abounds. A large number of silicates would be of great value if found in vast quantities. The same also is true of the metals. Platinum, considering its properties, would be the most valuable metal for the most varied technical applications, did it only occur as abundantly as iron. Hence also it follows that our idea of the value of a mineral does not always express its utility. Limiting ourselves to well known species, we find single minerals of the first group, the metalloids, of very general utility; of this, sulphur is an illustration. Even the rare variety of carbon, the diamond, is indispensable in glass-working; and on its employment as an ornamental gem is based a special branch of art, namely, diamond grinding, which is carried on in England and Holland. While the diamond is the hardest of all bodies, so its working is the most difficult. The hardness of the diamond exceeds that of other minerals so much, that no substance but its own dust can be used in grinding it. In grinding diamonds horizontal metal disks are used, which are made to rotate with immense velocity by means of a lathe. The diamond to be ground is cemented fast to a stem of wood or brass. The metal wheel is coated with oil, and strewn with the powder of impure or very small diamonds. Even the chips and fragments of the larger diamonds obtained are brought into use for the same purpose by pounding them in a steel mortar. After the wheel has been properly adjusted, the diamond is so fixed by means of the stem to which it has been cemented, that a certain part to be ground shall press upon the wheel, which is then to be set in motion. In this operation the diamond powder becomes finer, and the diamond is worn away by the attrition. After one face has been properly ground, the diamond is fixed in another position, and the operation thus repeated until the cutting is completed. Simple contrivances are adopted for so holding the diamond, with its stem, that it may be ground at any angle required.
The forms produced by grinding the diamond are the brilliant and the rosette or rose (pl. 32, figs. 85, 87, 90, 84, and 91). The object of giving so many facets to the diamond is to increase its sparkling, the light being reflected in all directions from the great number of variously inclined faces.
The groups which succeed the metalloids, as the native metals, the tellurids, antimonids, arsenids, sulphurets, and selenids, are all valuable on account of the metals to be obtained from them. The native metals are mixtures of various metallic substances. Native gold, for instance, always contains silver; native platinum almost always includes with it iridium, osmium, rhodium, palladium, ruthenium, iron, and manganese. The other groups contain ores of metals. A metal is found in chemical combination with a metalloid, as sulphur, selenium, tellurium, arsenic, and antimony. Economical purposes, therefore, require that the native metals, which are nearly all of the precious class, be separated from these various mixtures, and that all the components be preserved. In the other classes, on the other hand, the metal is generally preserved at the expense of the metalloid, which is mostly lost. All of these, excepting silver, belong to the baser metals. The separation of the noble metals is generally effected by solution and precipitation of the individual species from the solution. The manufacture of silver, and of the baser metals, is effected at the mining establishment, and depends principally on the separation of those substances which give to the ore its distinguishing feature. Thus from the arsenids we obtain the combined metal, silver for instance, by roasting the ore: this drives off the arsenic in the form of vapor of arsenious acid. Sulphur is dissipated in the same way; and the processes by which a separation from injurious combinations is effected, are sometimes so complicated as to form a considerable portion of the occupation of the workers in metals.
Of the oxydes some are important as being ores from which their metals may be economically obtained. Thus iron is obtained from its oxydes, or from minerals rich in this latter substance. Not less important is silica, in its different varieties. All our different forms of glass are combinations of silica with powerful bases, especially with potassa and soda. By the combination of impure carbonate of potassa, impure silica, and a certain quantity of lime, we obtain the well known green bottle and window glass. White window glass is obtained in the same way, by the addition of some decolorizing agent which shall remove the green color produced by the presence of protoxyde of iron. Peroxyde of manganese is used for this purpose, as by it the protoxyde of iron may be changed into peroxyde: this, when in large quantity, gives the glass a yellow color, but in less amount scarcely tinges it at all. For the better sorts of glass, only the purest materials are used, the silica, for instance, being furnished by the quartz rock, which contains it in great purity. By the addition of oxyde of lead the glass becomes more fluid, and such easily melted glass is used for preparing the colors for glass and porcelain painting, by melting with it various metallic oxydes. Thus, by the addition of oxyde of cobalt we get a dark blue glass, by that of oxyde of copper one of a beautiful reddish brown, which is pounded and rubbed to a fine powder. This powder, mixed with a drying oil, is applied to the glass or porcelain surface to be painted. After the paint has somewhat dried, the objects to which it is applied are exposed to a red heat, which melts the glass into an enamel.
Besides the application of silica in the manufacture of glass, no less important is the use made of several of its natural combinations. Pore elain is obtained by strongly heating together alumina, an alkali, and silica; for this purpose a substance of native occurrence is employed, namely, the kaolin or porcelain earth, which is found in large quantities at various localities. Perfectly clear and colorless rock crystal is used for purposes of ornament. Some colored varieties of silica, as the amethyst, are also applied to the same purpose. Chalcedony and agates also consist essentially of silica, handsomely colored by various oxydes; they furnish an excellent material for many instruments on account of their great hardness. Thus, plates of agate are used to suspend the knife-edges of delicate balances; the pivot holes of magnetic needles are also made of this same material, to avoid undue friction. Valuable dishes and capsules, mortars for chemical purposes, and many other useful implements of art, are constructed of these substances.
Some of the silicates constitute minerals of great value. The most important are those belonging to the family of feldspar. Feldspar is an essential ingredient of entire rocks, as granite and gneiss; minerals allied to this are important constituents also of other rocks, as labradorite and syenite. Feldspathic minerals, as constituents of the soil, are of the utmost value. Most of them disintegrate readily under atmospheric influences, and yield up to the plants in the soil those inorganic ingredients so necessary to their growth.
On examining certain minerals, as granites, which are rich in feldspar, we shall find that they gradually lose their compactness by exposure to the weather, crumbling into a fine sand. On investigation this is found to be rich in alumina and poor in alkali. In all probability it is the mechanical agency of the water, with the unceasing alternations of temperature, together with the dissolving action of the carbonic acid contained in the water, that wears away the stone; the carbonic acid also extracts the stronger bases, and thus contributes, in a great measure, to the disintegration of the rock. It is also to be observed, that the silica, in the condition in which it is separated from alkaline silicates, by aqueous influence, is very soluble; also, that the feldspar, with its alkalies, communicates to water a proportion of that dissolved silica so necessary to the sustenance of certain plants, especially the grasses, and which is deposited in their tissues. It has already been mentioned that purer beds of feldspar furnish, in many localities, by their decomposition, masses of the finer porcelain earth or kaolin. And it is not only this naturally-formed kaolin that is made use of in the manufacture of different kinds of porcelain and stone-ware; the mixtures of the constituents must frequently be varied. Thus, in the manufacture of porcelain, the coarser qualities of feldspar are ground, and mixed up with the finer porcelain earth, in the proportions required for the particular kind of ware.
The class of the salts is no less important. Carbonate of lime, in the form of limestone, furnishes an excellent building material, and is one of the most important constituents of a fertile soil. Besides this general utility of carbonate of lime, certain varieties, forming entire rocks, find valuable applications. The granular crystalline form of compact carbonate of lime is known as marble, the different varieties being produced by the fineness of the grains, or the varying character and distribution of the coloring matters. The coarse blocks of marble, after being extracted from the quarries, are taken to marble works, and there cut up into slabs and other forms, by means of broad saw-like steel blades, stretched in frames. The lithographic stone, used so extensively at the present time, is also a carbonate of lime. Chalk is another of these carbonates of lime, differing, however, from the rest mentioned, in consisting largely of the calcareous shells of minute infusoria and other animals, thus more nearly related to the muschelkalk. Many useful applications of carbonate of lime depend on the circumstance that the carbonic acid is driven off at a strong red heat, the lime retaking the acid from the atmosphere when exposed to the latter. Burnt or quick lime, when water is poured over it, takes up a certain amount, forming a definite chemical combination, a hydrate of lime, which, with an additional quantity of water, forms mortar. The hydrate loses its water in the air, passing slowly and by, degrees into a carbonate again, and thus hardens into a mass resembling the original limestone in compactness.
Sulphate of lime is also used for many purposes. In the form of karstenite, or anhydrite, it is used as a building material. Anhydrite becomes gradually converted into gypsum by the action of the atmosphere, gypsum being a combination of water and sulphate of lime. In the native mass gypsum is known as alabaster, and on account of its softness may be applied to various purposes.
The vitriols are used in coloring. Green vitriol, with tannic acid, furnishes the black color, ink; blue vitriol is used for similar purposes; a third salt of sulphuric acid, used in dyeing, is alum. Its value, like that of most salts of alumina, consists in its being a good mordant, that is, it forms permanent combinations with the fibre of many fabrics; the color being firmly combined with the alum, is indissolubly united to the cloth. The fabric intended to be dyed may be first steeped in the alum solution, before the application of the coloring matter, or both may be applied together. The uses of rock salt, or common cooking salt, are well known. Saltpetre, as well that of soda as of potassa, is the single substance yielding nitric acid. This is distilled in glass retorts, with sulphuric acid, by which the nitric acid is expelled and driven over into large glass reservoirs. Nitric acid is indispensable in the separation of metals, as gold from silver; its uses in other departments of the arts are manifold. Vast quantities of saltpetre are used in the manufacture of gunpowder, this consisting of saltpetre, sulphur, and charcoal. It is also used to advantage in pickling and curing meat. Borax is valuable as a flux for metals, as also for purifying the metals from oxydes. It also furnishes various borates of importance. Borate of lead forms the medium by which gold is applied to porcelain in the operation of gilding this ware.
The alkaline sulphates are used both in the arts and in medicine. Sulphate of soda, obtained from salt works and sea water, as also in the manufacture of soda, constitutes what is known as Glauber Salts.
Errata
Possible errors in the original plates compared to their descriptions.
- Plate 35:
- Two figures labeled 32
- Fig. 31 not in the text